WO2009126577A2 - Pesticidal compositions - Google Patents
Pesticidal compositions Download PDFInfo
- Publication number
- WO2009126577A2 WO2009126577A2 PCT/US2009/039664 US2009039664W WO2009126577A2 WO 2009126577 A2 WO2009126577 A2 WO 2009126577A2 US 2009039664 W US2009039664 W US 2009039664W WO 2009126577 A2 WO2009126577 A2 WO 2009126577A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- alcohol
- active ingredient
- weight
- mineral oil
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N27/00—Biocides, pest repellants or attractants, or plant growth regulators containing hydrocarbons
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N31/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic oxygen or sulfur compounds
- A01N31/02—Acyclic compounds
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/02—Saturated carboxylic acids or thio analogues thereof; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/36—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a singly bound oxygen or sulfur atom attached to the same carbon skeleton, this oxygen or sulfur atom not being a member of a carboxylic group or of a thio analogue, or of a derivative thereof, e.g. hydroxy-carboxylic acids
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/24—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with two or more hetero atoms
- A01N43/32—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with two or more hetero atoms six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N61/00—Biocides, pest repellants or attractants, or plant growth regulators containing substances of unknown or undetermined composition, e.g. substances characterised only by the mode of action
- A01N61/02—Mineral oils; Tar oils; Tar; Distillates, extracts or conversion products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/02—Medicinal preparations containing materials or reaction products thereof with undetermined constitution from inanimate materials
- A61K35/04—Tars; Bitumens; Mineral oils; Ammonium bituminosulfonate
- A61K35/06—Mineral oils, e.g. paraffinic oils or aromatic oils based on aromatic hydrocarbons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11B—PRODUCING, e.g. BY PRESSING RAW MATERIALS OR BY EXTRACTION FROM WASTE MATERIALS, REFINING OR PRESERVING FATS, FATTY SUBSTANCES, e.g. LANOLIN, FATTY OILS OR WAXES; ESSENTIAL OILS; PERFUMES
- C11B9/00—Essential oils; Perfumes
Definitions
- the present disclosure relates to pesticidal compositions and a method of controlling insects and other pests. More particularly, the disclosure relates to pesticidal compositions containing mineral oil and one or more additional components which, when used in combination, act synergistically to control pests such as arthropods.
- insects and other arthropod pests can have negative effects on the quality of human life. For instance, when found in the home, insects and other arthropods can be a source of annoyance due purely to their presence. They may also spread disease and allergens. Additionally, when found on plants and crops, insects and other pest arthropods can destroy foliage and fruit, and may adversely affect plant and crop growth, quality, and yield.
- insects which are particularly undesirable are domestic cockroaches. These insects produce allergens in their saliva, fecal material, cast skins and body parts which may be particularly problematic for people suffering from allergies.
- cockroaches It is domestically and commercially desirable to control cockroaches through the use of pesticide products. It is also desirable for to control other crawling arthropods, such as ants, beetles, earwigs, silverfish, crickets, spiders, centipedes and various flying insects including flies, mosquitoes, gnats, moths, wasps, hornets, bees, and the like.
- crawling arthropods such as ants, beetles, earwigs, silverfish, crickets, spiders, centipedes and various flying insects including flies, mosquitoes, gnats, moths, wasps, hornets, bees, and the like.
- the present disclosure relates to pesticidal compositions and a method of controlling pests such as arthropods and, in particular, insects.
- the pesticidal compositions of embodiments of the present disclosure are particularly effective at controlling cockroaches. More particularly, the disclosure relates to a pesticidal composition containing mineral oil and one or more additional components which, when used in combination, act synergistically to control insect and other pest populations.
- the present disclosure is directed to a pesticidal composition
- a pesticidal composition comprising a first active ingredient and a second active ingredient.
- the first active ingredient is a mineral oil and the second active ingredient is an ester compound.
- the ester compound is selected from the group consisting of ethyl lactate, ⁇ -butyrolactone, triacetin and combinations thereof.
- the pesticidal composition comprises a first active ingredient and a second active ingredient.
- the first active ingredient is a mineral oil and the second active ingredient is an alcohol selected from the group consisting of methanol, 1-proponal, 1-butanol, 1-hexanol, 1-heptanol, 1-octanol, 1- nonanol, 1-decanol, 1-undecanol, 1-dodecanol, 1-tetradecanol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, 2-ethoxyethanol and combinations thereof.
- the pesticidal composition comprises a first active ingredient and a second active ingredient.
- the first active ingredient is a mineral oil and the second active ingredient is selected from the group consisting of hexylene glycol, dioxane, nitromethane, acetophenone, pyridine and combinations thereof.
- the present disclosure is directed to a method of controlling arthropods.
- An arthropod is contacted with a pesticidally effective amount of a composition comprising a first active ingredient and a second active ingredient.
- the first active ingredient is a mineral oil and the second active ingredient is an ester compound.
- an arthropod is contacted with a pesticidally effective amount of a composition comprising a first active ingredient and a second active ingredient.
- the first active ingredient is a mineral oil and the second active ingredient is an alcohol selected from the group consisting of methanol, 1- proponal, 1-butanol, 1-hexanol, 1-heptanol, 1-octanol, 1-nonanol, 1-decanol, 1- undecanol, 1-dodecanol, 1-tetradecanol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, 2-ethoxyethanol and combinations thereof.
- the arthropod is contacted with a pesticidally effective amount of a composition comprising a first active ingredient and a second active ingredient.
- the first active ingredient is a mineral oil and the second active ingredient is selected from the group consisting of hexylene glycol, dioxane, d-limonene, nitromethane, acetophenone, pyridine and combinations thereof.
- a further aspect of the present disclosure is directed to a method of controlling cockroaches.
- a cockroach is contacted with a pesticidally effective amount of a composition comprising a first active ingredient and at least 3% of a second active ingredient by weight of the composition.
- the first active ingredient is a mineral oil and the second active ingredient is an alcohol.
- Figure 1 is a chart depicting the effects of compositions comprising mineral oil and/or ethyl lactate on the mortality of adult German cockroaches as discussed in Example 1.
- Figure 2 is a chart depicting the effects of compositions comprising mineral oil and/or butyl lactate on the mortality of adult German cockroaches as discussed in Example 2.
- compositions comprising mineral oil in combination with ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-C 14 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, ⁇ -butyrolactone, nitromethane, acetophenone, triacetin, pyridine, 2-ethoxyethanol or combinations thereof are more effective at killing targeted insects and pests than compositions comprising only mineral oil or compositions comprising ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-C 14 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, iso
- the present disclosure is directed to a pesticidal composition
- a pesticidal composition comprising a first active ingredient and a second active ingredient, the first active ingredient being mineral oil, and the second active ingredient being selected from the group consisting of ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-C 14 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, ⁇ -butyrolactone, nitromethane, acetophenone, triacetin, pyridine, 2-ethoxyethanol and combinations thereof.
- the composition may be sprayed or otherwise applied directly on crawling or flying insects or other arthropod pests in sufficient concentrations to kill the pests.
- the pesticidal compositions of the present disclosure comprise a first active ingredient which is mineral oil.
- a first active ingredient which is mineral oil.
- mineral oil is PD-23 (available from Chemtura Corporation, Middlebury, CT).
- PD-23 is a light grade petroleum distillate having a flash point of 230 0 F (HO 0 C), as measured by ASTM D-92, and a vapor pressure of less than 0.1 mmHg at 20 0 C.
- PD- 23 is a highly refined petroleum distillate that physically resembles a very light, clear oil, and is made by fractionating oil to a narrow boiling range, then catalytically hydrotreating it to remove all aromatic and other unsaturated hydrocarbons.
- PD-23 is hydrophobic, colorless, tasteless, virtually odorless, and color fast.
- PD-23 generally has a low viscosity, e.g., about 2.6 centistokes (cSt) at 40 0 C or a Saybolt Universal Seconds (SUS) viscosity of about 34 SUS at 100 0 F (38 0 C).
- Suitable mineral oils may also be used including, for example, PD-25 and PD-28 (both available from Chemtura Corporation, Middlebury, CT).
- PD-25 typically has a viscosity of about 3.5 cSt at 40 0 C or about 39 SUS at 100 0 F (38 0 C).
- PD-28 typically has a viscosity of about 4.2 cSt at 40 0 C or about 39 SUS at 100 0 F (38 0 C).
- Another suitable mineral oil is Exxsol D95 (available from Exxon Mobil).
- Exxsol D95 is a de-aromatized petroleum distillate.
- Exxsol D95 typically has a viscosity of about 1.92 cSt at 40 0 C.
- the viscosity of the mineral oil will range from about 2.0 cSt at 40 0 C to about 5.0 cSt at 40 0 C, and more typically will be from about 2.6 cSt at 40 0 C to about 4.2 cSt at 40 0 C. In other embodiments the viscosity of the mineral oil is from about 1.5 cSt to about 5.0 cSt at 40 0 C. In general, it has been determined that pesticidal compositions of the present disclosure that comprise a mineral oil having a relatively low viscosity are more effective at killing pests than pesticidal compositions comprising mineral oil having a higher viscosity.
- the pesticidal composition comprises more than one mineral oil.
- the total amount of mineral oil present in the pesticidal composition may be at least about 5% (by weight of the composition), at least about 10% (by weight of the composition), at least about 20% (by weight of the composition), at least about 30% (by weight of the composition), at least about 40% (by weight of the composition), or even at least about 50% (by weight of the composition).
- the pesticidal compositions will comprise from about 5% (by weight of the composition) to about 90% (by weight of the composition) of mineral oil, and more typically from about 40% (by weight of the composition) to about 80% (by weight of the composition) of mineral oil.
- the mineral oil is a petroleum distillate composition.
- the amount of petroleum distillate composition in the pesticidal composition may generally be the same as the amounts listed for the mineral oil as described above.
- the pesticidal compositions of the present disclosure further comprise at least one second active ingredient selected from the group consisting of ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-Cl 4 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2- phenylethanol, diacetone alcohol, ⁇ -butyrolactone, nitromethane, acetophenone, triacetin, pyridine, 2-ethoxyethanol and combinations thereof.
- at least one second active ingredient selected from the group consisting of ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-Cl 4 saturated straight-chain alcohol, isopropyl alcohol, 2-but
- compositions comprising a combination of mineral oil and one or more of ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-C 14 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol and diacetone alcohol, ⁇ - butyrolactone, nitromethane, acetophenone, triacetin, pyridine, 2-ethoxyethanol, are substantially more effective in killing insects than either mineral oil in the absence of one of these second active ingredients, or any of the second active ingredients in the absence of mineral oil.
- the pesticidal composition comprises more than one second active ingredient.
- the additional second active ingredient or ingredients may generally be selected from the list of compounds described above or below as potential second active ingredients.
- the total amount of second active ingredient or ingredients present in the pesticidal composition may be at least about 3% (by weight of the composition), at least about 5% (by weight of the composition), at least about 10% (by weight of the composition), at least about 20% (by weight of the composition), at least about 30% (by weight of the composition), at least about 40% (by weight of the composition), or even at least about 50% (by weight of the composition).
- the total amount of second active ingredient present in the pesticidal compositions will typically be from about 3% (by weight of the composition) to about 95% (by weight of the composition), more typically from about 5% (by weight of the composition) to about 95% (by weight of the composition), and more preferably from about 10% (by weight of the composition) to about 60% (by weight of the composition).
- the amount of each particular second active ingredient present in the composition may vary depending on the ingredient, the amount of mineral oil in the composition, and whether or not more than one second active ingredient is present in the composition.
- the second active ingredient is selected from ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene and combinations thereof. In another embodiment, the second active ingredient is selected from ethyl lactate, butyl lactate, isopropyl myristate, d-limonene and combinations thereof.
- the second active ingredient is selected from the group consisting of ethyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene and combinations thereof and, in a further embodiment, ethyl lactate, isopropyl myristate, d-limonene and combinations thereof.
- ethyl lactate may be present in the composition in an amount of from about 5% (by weight of the composition) to about 95% (by weight of the composition), and more preferably from about 10% (by weight of the composition) to about 60% (by weight of the composition).
- compositions comprising about 12.5% (by weight of the composition) mineral oil and about 37.5% (by weight of the composition) ethyl lactate, or about 25% (by weight of the composition) mineral oil and about 25% (by weight of the composition) ethyl lactate, or about 37.5% (by weight of the composition) mineral oil and about 12.5% (by weight of the composition) ethyl lactate, or about 5% (by weight of the composition) mineral oil and about 45% (by weight of the composition) ethyl lactate have been found to be particularly effective at killing insects such as cockroaches.
- Butyl lactate may be present in the composition in an amount of from about 5% (by weight of the composition) to about 90% (by weight of the composition), more preferably from about 10% (by weight of the composition) to about 80% (by weight of the composition), and more preferably from about 10% (by weight of the composition) to about 60% (by weight of the composition).
- compositions comprising about 12.5% (by weight of the composition) mineral oil and about 37.5% (by weight of the composition) butyl lactate, or about 25% (by weight of the composition) mineral oil and about 25% (by weight of the composition) butyl lactate, or about 37.5% (by weight of the composition) mineral oil and about 12.5% (by weight of the composition) butyl lactate have been found to be particularly effective at killing insects such as cockroaches.
- Isopropyl myristate may be present in the composition in an amount of from about 5% (by weight of the composition) to about 95% (by weight of the composition), more preferably from about 10% (by weight of the composition) to about 80% (by weight of the composition), and more preferably from about 10% (by weight of the composition) to about 60% (by weight of the composition).
- compositions comprising about 60% (by weight of the composition) mineral oil and about 20% (by weight of the composition) isopropyl myristate, or about 50% (by weight of the composition) mineral oil and about 30% (by weight of the composition) isopropyl myristate, or about 40% (by weight of the composition) mineral oil and about 40% (by weight of the composition) isopropyl myristate have been found to be particularly effective at killing insects such as cockroaches.
- Hexylene glycol may be present in the composition in an amount of from about 5% (by weight of the composition) to about 90% (by weight of the composition), and more preferably from about 10% (by weight of the composition) to about 60% (by weight of the composition).
- Compositions comprising about 60% (by weight of the composition) mineral oil and about 20% (by weight of the composition) hexylene glycol have been found to be particularly effective at killing insects such as cockroaches.
- Dioxane may be present in the composition in an amount of from about 5% (by weight of the composition) to about 80% (by weight of the composition), more preferably from about 5% (by weight of the composition) to about 60% (by weight of the composition), and more preferably from about 10% (by weight of the composition) to about 60% (by weight of the composition).
- Compositions comprising about 40% (by weight of the composition) mineral oil and about 40% (by weight of the composition) dioxane have been found to be particularly effective at killing insects such as cockroaches.
- D-limonene may be present in the composition in an amount of from about 3% (by weight of the composition) to about 90% (by weight of the composition), and more preferably from about 5% (by weight of the composition) to about 90% (by weight of the composition), and more preferably from about 5% (by weight of the composition) to about 60% (by weight of the composition).
- Compositions comprising about 40% (by weight of the composition) mineral oil and about 40% (by weight of the composition) d-limonene have been found to be particularly effective at killing insects such as cockroaches.
- the second active ingredient is an ester compound.
- ester compounds When ester compounds are present in the pesticidal composition as a second active ingredient, additional second active ingredients that are not ester compounds may also be present in the composition.
- the amount of ester compounds that are present in the pesticidal composition may be at least about 50% of all the second active ingredients, at least about 65% of all the second active ingredients, at least about 75% of all the second active ingredients, at least about 85% of all the second active ingredients, at least about 95% of all the second active ingredients, or even at least about 99% of all the second active ingredients.
- the total amount (i.e., about 100%) of second active ingredients present in the pesticidal composition is ester compounds.
- the ester compounds present in the pesticidal composition may be selected from the group consisting of ethyl lactate, butyl lactate, isopropyl myristate, ⁇ -butyrolactone, triacetin and combinations thereof. These listed esters may be present in the pesticidal composition in an amount of at least about 1% by weight of the composition, at least about 3%, at least about 5%, at least about 10% or even at least about 15% by weight of the composition. In one embodiment, the ester compounds are present in an amount from about 10% to about 60% by weight of the composition.
- the ester compounds are selected from the group consisting of ethyl lactate, butyl lactate, isopropyl myristate and combinations thereof. In yet another embodiment, the ester compounds are selected from the group consisting of ethyl lactate, ⁇ -butyrolactone, triacetin and combinations thereof. In yet another aspect, the ester compounds are selected from the group consisting of ethyl lactate, ⁇ -butyrolactone, triacetin and combinations thereof.
- the second active ingredient is an alcohol.
- the pesticidal composition may include one alcohol compound or more than one alcohol compound without departing from the scope of the present disclosure.
- additional second active ingredients that are not alcohols may also be present in the composition.
- the amount of alcohol present in the pesticidal composition may be at least about 50% of all the second active ingredients, at least about 65% of all the second active ingredients, at least about 75% of all the second active ingredients, at least about 85% of all the second active ingredients, at least about 95% of all the second active ingredients, or even at least about 99% of all the second active ingredients.
- the total amount (i.e., about 100%) of second active ingredients present in the pesticidal composition is an alcohol or alcohols.
- the alcohol utilized in the second active ingredient may be selected from the group consisting of Cl -C 14 saturated straight-chain alcohols, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, 2-ethoxyethanol and combinations thereof. These alcohols may be present in the pesticidal composition in at least about 1%, at least about 3%, at least about 5%, at least about 10% or even at least about 15% by weight of the composition. In one embodiment, the alcohols are present in an amount from about 10% to about 60% by weight of the composition.
- the alcohol is selected from the group consisting of methanol, 1-proponal, 1-butanol, 1-hexanol, 1-heptanol, 1-octanol, 1- nonanol, 1-decanol, 1-undecanol, 1-dodecanol, 1-tetradecanol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, 2-ethoxyethanol and combinations thereof.
- the alcohol is selected from methanol, 1-proponal, 1-butanol, 1-hexanol, 1-heptanol, 1-octanol, 1- nonanol, 1-decanol, 1-undecanol, 1-dodecanol, 1-tetradecanol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol and combinations thereof.
- the alcohol may also be selected from the group consisting of 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2- phenylethanol, diacetone alcohol, 2-ethoxyethanol and combinations thereof.
- the alcohol is selected from the group consisting of Cl -C 14 saturated straight-chain alcohols, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, 2-ethoxyethanol and combinations thereof.
- the pesticidal composition may include a plurality of alcohol compounds including a plurality of Cl -C 14 saturated straight chain alcohols.
- the Cl -C 14 saturated straight-chain alcohol is selected from the group consisting of methanol, ethanol, 1-proponal, 1-butanol, 1-hexanol, 1-heptanol, 1- octanol, 1-nonanol, 1-decanol, 1-undecanol, 1-dodecanol, 1-tetradecanol and combinations thereof.
- the alcohol is a C1-C9 saturated straight- chain alcohol and, in another embodiment, is a C1-C6 straight chain alcohol. In one embodiment, the alcohol is a C3-C7 saturated straight-chain alcohol.
- mineral oil and a second active ingredient selected from the group consisting of Cl -C 14 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2- butoxyethanol, 2-phenylethanol or diacetone alcohol may be present in the composition in a ratio based on the LDio or LD50 value of the alcohol, which optimizes the biological effect of the blend.
- the LDio of each of the active ingredients is determined, for instance, by the methods described in Example 9. A ratio is then determined based on about half of the LDio for mineral oil and about the LDi 0 of the second active ingredient. For example, as detailed in Example 9, the LDio for PD23 mineral oil was determined to be 0.754 mg/insect and the LDio for diacetone alcohol was determined to be 0.255 mg/insect.
- a ratio corresponding to the above amounts is 4 parts PD23 to 3 parts diacetone alcohol, or 4:3.
- a composition of this particular embodiment may comprise PD23 and diacetone alcohol, among other things, in a ratio of 4:3.
- those active ingredients may be present in the composition in a ratio of about 4:4.5 or about 1 :1.
- the ratio is determined based on the LDio for 1-propanol and PD23, respectively, of 0.472 mg/insect and 0.754 mg/insect.
- About half the LDio of PD23 is about 0.4 mg, and the LDi 0 of 1-propanol is about 0.45 mg, giving a ratio of about 4:4.5, or about 1 :1.
- the ratio may be determined as above, but based on the LDio:LDio of the mineral oil and second active ingredient. So, for example, in a composition comprising PD23 and 1-propanol, the PD23 and 1-propanol are present in a ratio of 8:4.5, or about 2:1.
- a Cl-C 14 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2- phenylethanol or diacetone alcohol may be present in the composition in a ratio relative to the mineral oil in the composition, where this ratio is determined based on the LD50 for the alcohol and the LD50 of the mineral oil. As above with respect to ratios based on LDio, such ratios may be determined based on LDso ⁇ LDso; or may alternatively be determined based on 0.5 LD50 for mineral oil and LD50 for the second active ingredient.
- mineral oil and a second active ingredient selected from the group consisting of Cl -C 14 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2- butoxyethanol, 2-phenylethanol, diacetone alcohol, 2-ethoxyethanol or combinations thereof may be present in the composition in a ratio which may be determined based
- the second active ingredient is a compound selected from the group consisting of hexylene glycol, dioxane, d-limonene, nitromethane, acetophenone, pyridine and combinations thereof. These listed compounds may be included with other second active ingredients. The amount of the listed compounds in the pesticidal composition may be at least about 50% of all the second active ingredients, at least about 65% of all the second active ingredients, at least about 75% of all the second active ingredients, at least about 85% of all the second active ingredients, at least about 95% of all the second active ingredients, or even at least about 99% of all the second active ingredients.
- the total amount (i.e., about 100%) of second active ingredients present in the pesticidal composition is compounds selected from hexylene glycol, dioxane, d-limonene, nitromethane, acetophenone, pyridine and combinations thereof.
- the second active ingredient is selected from the group consisting of hexylene glycol, dioxane, d-limonene, nitromethane, acetophenone, pyridine and combinations thereof.
- the second active ingredient is selected from hexylene glycol, dioxane, d-limonene and combinations thereof. In a further embodiment, the second active ingredient is selected from nitromethane, acetophenone, pyridine and combinations thereof.
- These compounds may be present in the pesticidal composition in at least about 1%, at least about 3%, at least about 5%, at least about 10% or even at least about 15% by weight of the composition. In one embodiment, the listed compounds are present in an amount from about 10% to about 60% by weight of the composition.
- the pesticidal compositions may comprise mineral oil plus two or more of the second active ingredients.
- the pesticidal composition comprises mineral oil, ethyl lactate, and isopropyl myristate.
- the pesticidal composition preferably comprises from about 5% (by weight of the composition) to about 30% (by weight of the composition) of ethyl lactate, and more typically from about 5% (by weight of the composition) to about 15% (by weight of the composition) of ethyl lactate; from about 5% (by weight of the composition) to about 60% (by weight of the composition) of isopropyl myristate, and more typically from about 5% (by weight of the composition) to about 30% (by weight of the composition) of isopropyl myristate; and from about 20% (by weight of the composition) to about 70% (by weight of the composition) of mineral oil.
- the pesticidal composition comprises mineral oil, d-limonene, and ethyl lactate.
- the pesticidal composition preferably comprises from about 5% (by weight of the composition) to about 60% (by weight of the composition) of d-limonene, from about 5% (by weight of the composition) to about 30% (by weight of the composition) of ethyl lactate, and from about 20% (by weight of the composition) to about 90% (by weight of the composition) of mineral oil.
- the pesticidal composition comprises mineral oil, d-limonene, and butyl lactate.
- the pesticidal composition preferably comprises from about 5% (by weight of the composition) to about 20% (by weight of the composition) of d-limonene, from about 5% (by weight of the composition) to about 20% (by weight of the composition) of butyl lactate, and from about 30% (by weight of the composition) to about 80% (by weight of the composition) of mineral oil.
- the pesticidal composition may comprise mineral oil, d-limonene, ethyl lactate, and isopropyl myristate.
- the pesticidal composition preferably comprises from about 5% (by weight of the composition) to about 20% (by weight of the composition) of d- limonene, from about 5% (by weight of the composition) to about 20% (by weight of the composition) of ethyl lactate, from about 20% (by weight of the composition) to about 40% (by weight of the composition) of isopropyl myristate, and from about 30% (by weight of the composition) to about 80% (by weight of the composition) of mineral oil.
- the pesticidal compositions of the present disclosure may further comprise a plant essential oil or derivative thereof, which may also have pesticidal properties.
- plant essential oils or derivatives may be extracted from natural sources or synthetically made, and generally contain, as at least one major constituent, an acyclic monoterpene alcohol or aldehyde, a benzenoid aromatic compound containing at least one oxygenated substituent or side chain, or a monocarbocyclic terpene generally having a six membered ring bearing one or more oxygenated substituents.
- oils or their derivatives include, but are not limited to, members selected from the group consisting of aldehyde C 16 (pure), alpha-terpineol, amyl cinnamic aldehyde, amyl salicylate, anisic aldehyde, benzyl alcohol, benzyl acetate, cinnamaldehyde, cinnamic alcohol, carvacrol, carveol, citral, citronellal, citronellol, dimethyl salicylate, eucalyptol (cineole), eugenol, iso-eugenol, galaxolide, geraniol, guaiacol, ionone, menthol, methyl anthranilate, methyl ionone, methyl salicylate, nerol, alpha-phellandrene, pennyroyal oil, perillaldehyde, 1- or 2-phenyl ethyl alcohol
- suitable oils include, for example, castor oil, cedar oil, cinnamon and cinnamon oil, citronella and citronella oil, cloves and clove oil, corn oil, cottonseed oil, garlic and garlic oil, geranium oil, lemongrass oil, linseed oil, mint and mint oil, peppermint and peppermint oil, rosemary and rosemary oil, sesame and sesame oil, soybean oil, white pepper, and the like.
- the plant essential oil or derivative is geraniol.
- the pesticidal compositions will typically comprise from about 1% (by weight of the composition) to about 60% (by weight of the composition of the plant essential oil or derivative, more typically from about 2% (by weight of the composition) to about 60% (by weight of the composition), and more typically from about 2% (by weight of the composition) to about 20% (by weight of the composition) of the plant essential oil or derivative.
- the pesticidal composition includes a first active ingredient that is a mineral oil, at least one second active ingredient that is selected from the group consisting of ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-Cl 4 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol, diacetone alcohol, and combinations thereof and a third active ingredient that is a plant essential oil or derivative.
- the plant essential oil or derivative is geraniol.
- the composition also includes nerol.
- the composition comprises from about 10% to about 70% of the first ingredient by weight of the composition, from about 5% to about 50% of the second ingredient by weight of the composition and from about 0.5% to about 15% of the third active ingredient by weight of the composition.
- the amount of geraniol present in the composition may be at least about 2% by weight of the composition, at least about 4% or at least about 6% by weight of the composition. In one embodiment, the amount of geraniol present in the composition is from about 0.5% to about 8% by weight of the composition. Nerol may be present in the composition in an amount from about 0.5% to about 3% by weight of the composition.
- the pesticidal composition comprises mineral oil, and at least two second active ingredients, each second active ingredient being selected from the group consisting of ethyl lactate, butyl lactate, isopropyl myristate, hexylene glycol, dioxane, d-limonene, a Cl-Cl 4 saturated straight-chain alcohol, isopropyl alcohol, 2-butanol, isobutyl alcohol, tertiary butyl alcohol, 2- butoxyethanol, 2-phenylethanol, diacetone alcohol, and combinations thereof and a third active ingredient that is a plant essential oil or derivative.
- the plant essential oil or derivative is geraniol.
- the composition also includes nerol.
- the pesticidal composition comprises a mineral oil, ethyl lactate, isopropyl myristate and geraniol.
- the composition may also include nerol.
- Additional additives may include essential oils such as lemongrass oil and carbon dioxide (described below).
- the pesticidal composition comprises, by weight of the composition, about 43% mineral oil, about 15% ethyl lactate, about 30% isopropyl myristate and about 6% geraniol.
- the composition may also include about 3% nerol and about 1% lemongrass oil.
- the pesticidal composition may further comprise other known insecticidal or pesticidal agents, including, for example, synergists such as piperonyl butoxide, MGK 264, and dillapiol; as well as other plant-derived insecticidal or pesticidal materials such as pyrethrum.
- synergists such as piperonyl butoxide, MGK 264, and dillapiol
- other plant-derived insecticidal or pesticidal materials such as pyrethrum.
- the composition may also comprise an amount of water.
- the composition contains water in an amount of, by weight of the composition, at least about 10%, at least about 25%, at least about 50% or even at least about 75%.
- the amount of water in the solution is from about 10% to about 50% by weight of the composition and, in another embodiment, from about 25% to about 75% by weight of the composition.
- compositions of the present disclosure may further comprise a suitable solvent, carrier or emulsif ⁇ er.
- suitable solvents or carriers include water, acetone, alcohols such as ethanol and isopropyl alcohol, dimethyl ether, and chlorinated hydrocarbon solvents such as methylene chloride and methyl chloroform.
- emulsifiers would include soaps (such as fatty acid soaps), cationic, ionic and non-ionic compounds.
- Additional carriers for use in various granular formulations include, for example, corn cob grits, diatomaceous earth, sand, clay, and the like.
- the pesticidal compositions of the present disclosure may be dispensed in any conventional manner, e.g., from a standard pump-spray container.
- the pesticide is in a ready-to-use form in which the composition does not need to be diluted before application.
- the pesticidal composition may be in a concentrate form. The end-user of the concentrate composition may dilute the concentrate prior to use by, for example, addition of water.
- the pesticidal composition is diluted in the pump spray bottle.
- the concentrate pesticidal composition may be diluted by addition of water in an amount of, by weight of the concentrate composition, at least about 40%, at least about 50%, at least about 75%, at least about 100%, at least about 150%, at least about 200% or even at least about 300%.
- the pesticidal composition may include emulsif ⁇ ers such as surfactants (e.g., surfactants such as fatty acid soaps).
- emulsif ⁇ ers such as surfactants (e.g., surfactants such as fatty acid soaps).
- Suitable compounds may be cationic, ionic, and non-ionic.
- Suitable emulsifying compounds include alkyl aryl sulionatc-based einulsifiers, polyoxycthylenet * and long-chain alcohols,
- the pesticidal composition is packaged in a pressurized container such as a conventional aerosol container or the like.
- the composition may be pressurized utilizing any suitable propellant.
- suitable propellants include expandable gases such as carbon dioxide, nitrogen, propane, n-butane, isobutane, blends of propane and butane, HFC- 152a (difluoroethane), HFC- 134a (1,1,1,2-tetrafluoroethane), and combinations thereof.
- any conventional technique may be used to charge the aerosol container with carbon dioxide or other expandable gas.
- the gasser-shaker method may be used. This method involves shaking or agitating the container and its contents as the carbon dioxide (or other gas) is forced into the container through a valve, with the rest of the composition having been introduced into the container prior to installation of the valve.
- the rate of injection of the gas depends on the valve orifice size, and the shaking time depends on the container size, the amount of liquid, and the degree of agitation, as well as the temperature.
- the pesticidal composition may be saturated with carbon dioxide prior to introduction into the container, and then pumped under pressure to a rotary undercap filler and filled as in a normal undercap operation, or pressured into containers already equipped with valves.
- Other suitable methods known in the art may also be used.
- Suitable propellants which may be used are known in the art and include, for example, dimethyl ether, methyl ethyl ether, nitrous oxide, and the like.
- the composition will comprise propellant in an amount of from about 2% (by weight of the composition) to about 90% (by weight of the composition), and more typically in an amount of from about 2% (by weight of the composition) to about 10% (by weight of the composition).
- the pesticidal compositions of the present disclosure may be sprayed or otherwise contacted with insects or other pests to effectively control insect or other pest populations.
- the present disclosure is directed to a method of controlling insects and pests. The method comprises contacting the insects and pests with a pesticidally effective amount of a composition of the present disclosure.
- an arthropod is contacted with the pesticidal composition in a pesticidally effective amount.
- a "pesticidally effective amount" of the composition includes amounts that repel the arthropod or other pest and may include, in another embodiment, amounts of the composition that kill the arthropod or other pest.
- the pesticidal compositions of embodiments of the present disclosure may be used to control arthropods and, in another embodiment, to control insects.
- the pesticidal composition is used to control cockroaches.
- Control of the pest may include contacting the pest with the pesticidal composition.
- the pest is contacted with the pesticidal composition by spraying the composition directly on the crawling arthropod, flying insect or other pest.
- the composition may be sprayed in a sufficient amount to cause death.
- the pesticidal composition may also be sprayed in locations at which the pest is likely to come into contact with the composition.
- the amount of the pesticidal composition required to kill insects or other pests and the time until pest death will vary depending on the composition components, the type of insect species and its life stage, and the like.
- the time required to kill a pest contacted with the pesticidal composition of the present disclosure will typically range from mere seconds to several hours.
- the pesticidal compositions of the present disclosure are particularly effective against cockroaches, including the species Periplaneta Americana (American cockroach), Blattella germanica (German cockroach), Blattella asahinai (Asian cockroach), and Blatta orientalis (Oriental cockroach).
- the pesticidal compositions may also be used against a wide variety of other crawling insects or other arthropods, such as ants, water bugs, silverfish, crickets, spiders, ticks, mites, sowbugs, pillbugs, beetles, earwigs, centipedes, and the like.
- water bugs refers to aquatic insects in the order Hemiptera.
- the pesticidal compositions are effective against various flying insects including flies, mosquitoes, gnats, moths, wasps, hornets, bees, and the like.
- the pesticidal compositions of the present disclosure generally result in at least 80% mortality, more preferably at least 95% mortality, and more preferably 100% mortality of the insects on contact, depending on the specific composition, the amount of composition applied to the insect and the insect species and life stage.
- the mineral oil used in the following examples was PD-23 (available from Chemtura Corporation, Middlebury, CT), which is a light grade petroleum distillate having a flash point of 230 0 F (HO 0 C), as measured by ASTM D-92, and a vapor pressure of less than 0.1 mmHg at 20 0 C.
- PD- 23 is a highly refined petroleum distillate that resembles a very clear oil, and is made by fractionating oil to a narrow boiling range, then catalytically hydrotreating it to remove all aromatic and other unsaturated hydrocarbons.
- PD-23 is hydrophobic, colorless, tasteless, virtually odorless, and color fast.
- compositions comprising various concentrations of mineral oil and/or ethyl lactate at killing German cockroaches was tested.
- compositions comprising mineral oil and/or ethyl lactate in the following concentrations were prepared: 100% (wt/wt) mineral oil/0% (wt/wt) ethyl lactate; 90% (wt/wt) mineral oil/10% (wt/wt) ethyl lactate; 75% (wt/wt) mineral oil/25% (wt/wt) ethyl lactate; 50% (wt/wt) mineral oil/50% (wt/wt) ethyl lactate; 25% (wt/wt) mineral oil/75% (wt/wt) ethyl lactate; 10% (wt/wt) mineral oil/90% (wt/wt) ethyl lactate; 0% (wt/wt) mineral oil/100% (wt/wt) ethyl lactate.
- the ethyl lactate was obtained from Vertech Biosolvents, Inc., Downers Grove, IL.
- each composition containing either mineral oil, ethyl lactate, or a combination of mineral oil and ethyl lactate was diluted by 50% by mixing the composition with acetone in a 1 :1 ratio (wt/wt) to reduce viscosity prior to application of the compositions to the cockroaches.
- the amount of mineral oil, ethyl lactate, and acetone present in the final composition (A-G) is set forth below in Table 1.
- Toxicity evaluations were performed on 7-14 day old adult male and female German cockroaches (SCJ Strain, S. C. Johnson & Son, Racine, WI).
- cockroaches were transferred to individual 100 x 20 mm polystyrene Petri dishes and anesthetized with a 15-25 second exposure to carbon dioxide.
- the inside edge of each Petri dish was lightly coated with mineral oil plus petroleum jelly in a 1 :3 (wt/wt) ratio to minimize escape.
- Anesthetized cockroaches were positioned with their ventral side up, and a 1 ⁇ l drop of the diluted test composition was applied to the area between the meso- and metathoracic legs using a Rainin L- 10, 10 ⁇ l capacity pipette (Rainin Instrument, LLC, Oakland, CA). Five replications of 12 male cockroaches were done for each test composition. Separate tests were done using female cockroaches.
- Results were evaluated at 24 hours following treatment. Cockroaches were scored as either alive (dorsal side up, active movement when abdomen prodded with dissecting probe) or moribund/dead (dorsal side up and no movement when abdomen prodded or ventral side up and insect unable to right itself).
- Results are shown in Figure 1 and Table 1 , which lists the number of dead cockroaches out of 60 total cockroaches for each test performed using the diluted test compositions (A-G).
- composition C-F containing both mineral oil and ethyl lactate
- composition G ethyl lactate
- compositions comprising various concentrations of mineral oil and/or butyl lactate at killing cockroaches was tested.
- compositions comprising mineral oil and/or butyl lactate in the following concentrations were prepared: 100% (wt/wt) mineral oil/0% (wt/wt) butyl lactate; 90% (wt/wt) mineral oil/ 10% (wt/wt) butyl lactate; 75% (wt/wt) mineral oil/25% (wt/wt) butyl lactate; 50% (wt/wt) mineral oil/50% (wt/wt) butyl lactate; 25% (wt/wt) mineral oil/75% (wt/wt) butyl lactate; 10% (wt/wt) mineral oil/90% (wt/wt) butyl lactate; 0% (wt/wt) mineral oil/ 100% (wt/wt) butyl lactate.
- the butyl lactate was obtained from Vertech Biosolvents, Inc., Downers Grove, IL.
- each composition containing either mineral oil, butyl lactate, or a combination of mineral oil and butyl lactate was diluted by 50% by mixing the composition with acetone in a 1 :1 ratio (wt/wt) to reduce viscosity prior to application of the composition to the cockroaches.
- the amount of mineral oil, butyl lactate, and acetone present in the final compositions (H-N) is set forth below in Table 2.
- composition H cockroaches treated with test composition J-L (containing both mineral oil and butyl lactate) had significantly higher mortality rates than cockroaches treated with only mineral oil (composition H) or only butyl lactate (composition N), indicating a synergistic effect when the combination of mineral oil and butyl lactate is used.
- compositions comprising various concentrations of mineral oil and/or hexylene glycol at killing cockroaches was tested.
- compositions comprising mineral oil, hexylene glycol, or a combination of mineral oil and hexylene glycol were prepared.
- the amount of mineral oil and/or hexylene glycol in each composition is set forth below in Table 3, with the remainder of each composition consisting of acetone.
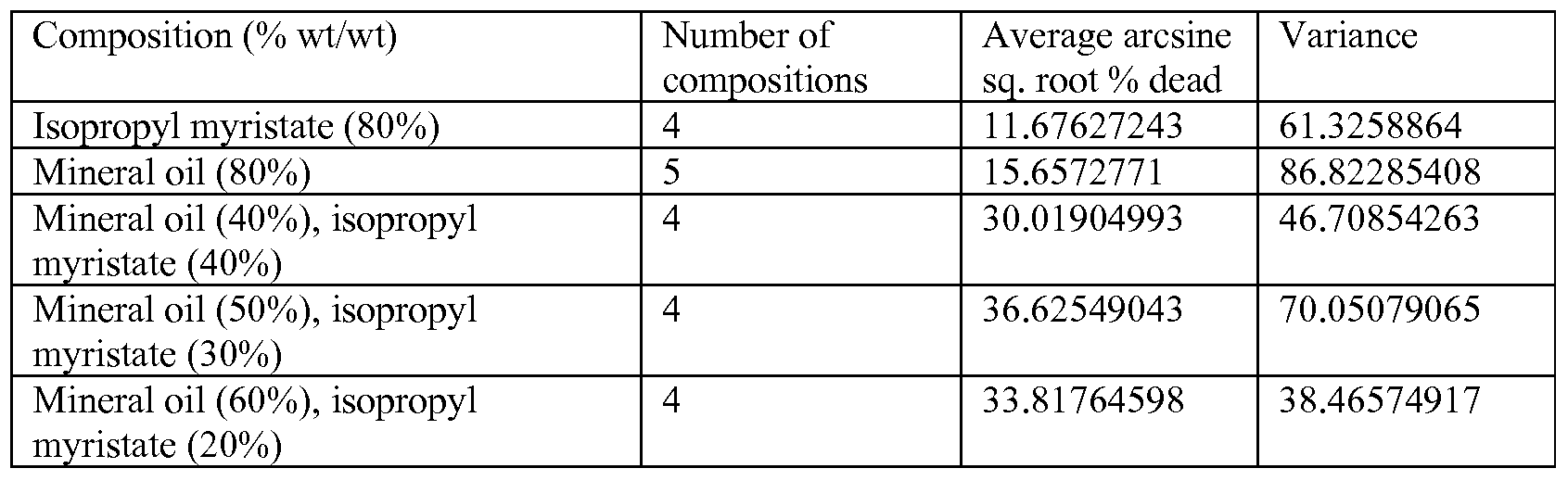
- Tests were generally performed as described in Example 1, except the number of cockroaches (all male) tested for each composition is set forth below in Table 3, and the compositions were not further diluted with acetone in a 1 :1 (wt/wt) ratio prior to testing. The results are shown in Table 3 below, which lists the number of cockroaches tested for each composition, the number of dead cockroaches out of the total number of cockroaches tested, the square root percent dead cockroaches, the proportion of dead cockroaches, and the arcsine square root percent dead cockroaches.
- compositions comprising various concentrations of mineral oil and/or isopropyl myristate at killing cockroaches was tested.
- compositions comprising mineral oil, isopropyl myristate, or a combination of mineral oil and isopropyl myristate were prepared.
- the amount of mineral oil and/or isopropyl myristate in each composition is set forth below in Table 4, with the remainder of each composition consisting of acetone.
- Tests were generally performed as described in Example 1, except the number of cockroaches (all male) tested for each composition is set forth below in Table 4, and the compositions were not further diluted with acetone in a 1 :1 (wt/wt) ratio prior to testing. The results are shown in Table 4 below, which lists the number of cockroaches tested for each composition, the number of dead cockroaches out of the total number of cockroaches tested, the square root percent dead cockroaches, the proportion of dead cockroaches, and the arcsine square root percent dead cockroaches. TABLE 4
- compositions comprising various concentrations of mineral oil and/or dioxane at killing cockroaches was tested.
- compositions comprising mineral oil, dioxane, or a combination of mineral oil and dioxane were prepared.
- the amount of mineral oil and/or dioxane in each composition is set forth below in Table 6, with the remainder of each composition consisting of acetone.
- Tests were generally performed as described in Example 1, except the number of cockroaches (all male) tested for each composition is set forth below in Table 6, and the compositions were not further diluted with acetone in a 1 :1 (wt/wt) ratio prior to testing. The results are shown in Table 6, which lists the number of cockroaches tested for each composition, the number of dead cockroaches out of the total number of cockroaches tested, the square root percent dead cockroaches, the proportion of dead cockroaches, and the arcsine square root percent dead cockroaches.
- compositions comprising various concentrations of mineral oil and/or d-limonene at killing cockroaches were tested.
- compositions comprising mineral oil, d-limonene, or a combination of mineral oil and d-limonene were prepared.
- the amount of mineral oil and/or d-limonene in each composition is set forth below in Table 7, with the remainder of each composition consisting of acetone.
- Tests were generally performed as described in Example 1, except the number of cockroaches (all male) tested for each composition is set forth below in Table 7, and the compositions were not further diluted with acetone in a 1 :1 (wt/wt) ratio prior to testing. The results are shown in Table 7, which lists the number of cockroaches tested for each composition, the number of dead cockroaches out of the total number of cockroaches tested, the square root percent dead cockroaches, the proportion of dead cockroaches, and the arcsine square root percent dead cockroaches.
- compositions comprising various types of mineral oil were tested.
- compositions comprising one of three different types of mineral oil in varying concentrations were prepared.
- the mineral oils used in this example were PD-23 (Sample No. 88446, 1997, available from Chemtura Corporation, Middlebury, CT), which is a low viscosity (e.g., about 2.6 cSt at 40 0 C or about 34 SUS at 100 0 F (38 0 C)) petroleum distillate; PD-25 (Sample No.
- Tests were generally performed as described in Example 1, except 2 ⁇ l of the compositions were applied to each cockroach, the number of cockroaches (all male) tested for each diluted composition is set forth below in Table 9, and the compositions were not further diluted with acetone in a 1 :1 (wt/wt) ratio prior to testing. The results are shown in Table 9. TABLE 9
- compositions containing the higher concentration of mineral oil were more effective against adult German cockroaches than the lower concentration compositions (i.e., the 20% compositions).
- the 20% compositions only the PD-23 had any effect against the cockroaches.
- an aerosol pesticidal composition of the present disclosure was prepared.
- the ingredients and amounts used to prepare the composition are set forth in Table 10 below.
- the composition was prepared by mixing the mineral oil, ethyl lactate, isopropyl myristate and geraniol together until homogenous.
- the resulting composition was placed in an aerosol can.
- the lid and actuator of the aerosol can were sealed onto the top of the can.
- the carbon dioxide was introduced into the can through the actuator to pressurize the can.
- the resulting product can be used as an aerosol spray pesticidal composition.
- PD23 and PD28 mineral oils were obtained from Sonneborn LLC, Mahwah, New Jersey. The physical properties of these oils are shown in Table 11. Each mineral oil was tested to determine the LD 50 and LDi 0 (Table 12).
- the alcohols tested comprise Cl -C 14 saturated straight chain alcohols, and alcohols selected from the group consisting of isopropyl alcohol, 2- butanol, isobutyl alcohol, tertiary butyl alcohol, 2-butoxyethanol, 2-phenylethanol and diacetone alcohol.
- the tested alcohols were obtained from Sigma-Aldrich Corp., St. Louis, Missouri. Each alcohol was tested to determine the LD50 and LD10 (Table 12).
- the dose-response mortality data were analyzed using probit analysis (PoloPlus ⁇ , LeOra Software Co., Petaluma CA, 2003) to estimate LD50 and LD10 values and their 95% confidence intervals. TABLE 12
- compositions comprising various alcohols and/or mineral oil at killing cockroaches was tested.
- compositions comprising alcohol and/or mineral oil are described in Table 13. The tests were performed as described in Example 1.
- an aerosol pesticidal composition of the present disclosure was prepared.
- the ingredients and amounts used to prepare the composition are set forth in Table 15 below.
- the composition was prepared by mixing a geraniol/nerol composition with the mineral oil, ethyl lactate, isopropyl myristate and lemongrass oil until homogenous.
- the resulting composition was placed in an aerosol can.
- the lid and actuator of the aerosol can were sealed onto the top of the can.
- the carbon dioxide was introduced into the can through the actuator to pressurize the can.
- the resulting product can be used as an aerosol pesticidal composition.
- compositions comprising various compounds and/or mineral oil at killing cockroaches was tested.
- the increase in mortality indicates a synergistic effect with the combination compared to the tested compound or mineral oil alone, with a higher difference indicating a greater synergy, ⁇ -butyrolactone , nitromethane, acetophenone, triacetin, pyridine and 2-ethoxyethanol demonstrated the greatest increase in mortality when combined with PD23 mineral oil, as compared to the other compounds tested.
Abstract
Description
Claims
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0911664-8A BRPI0911664A2 (en) | 2008-04-11 | 2009-04-06 | Pesticide composition and method for controlling arthropods and controlling cockroaches |
| EA201001616A EA018969B1 (en) | 2008-04-11 | 2009-04-06 | Pesticidal compositions |
| CA2720953A CA2720953A1 (en) | 2008-04-11 | 2009-04-06 | Pesticidal compositions |
| EP09730159A EP2271216A4 (en) | 2008-04-11 | 2009-04-06 | Pesticidal compositions |
| MX2010011081A MX2010011081A (en) | 2008-04-11 | 2009-04-06 | Pesticidal compositions. |
| AU2009233944A AU2009233944A1 (en) | 2008-04-11 | 2009-04-06 | Pesticidal compositions |
| JP2011504111A JP2011517685A (en) | 2008-04-11 | 2009-04-06 | Pesticide composition |
| CN2009801215262A CN102083311A (en) | 2008-04-11 | 2009-04-06 | Pesticidal compositions |
| IL208553A IL208553A0 (en) | 2008-04-11 | 2010-10-07 | Pesticidal compositions |
| MA33331A MA32292B1 (en) | 2008-04-11 | 2010-11-08 | Pesticide formulations |
| ZA2010/08005A ZA201008005B (en) | 2008-04-11 | 2010-11-09 | Pesticidal compositions |
Applications Claiming Priority (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US4429708P | 2008-04-11 | 2008-04-11 | |
| US61/044,297 | 2008-04-11 | ||
| US11523108P | 2008-11-17 | 2008-11-17 | |
| US61/115,231 | 2008-11-17 | ||
| US12/418,466 | 2009-04-03 | ||
| US12/418,466 US20090258889A1 (en) | 2008-04-11 | 2009-04-03 | Pesticidal compositions |
| US12/418,455 | 2009-04-03 | ||
| US12/418,455 US9005644B2 (en) | 2008-04-11 | 2009-04-03 | Pesticidal compositions |
| US12/418,443 US8231887B2 (en) | 2008-04-11 | 2009-04-03 | Pesticidal compositions |
| US12/418,443 | 2009-04-03 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2009126577A2 true WO2009126577A2 (en) | 2009-10-15 |
| WO2009126577A3 WO2009126577A3 (en) | 2010-04-22 |
Family
ID=41164174
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2009/039664 WO2009126577A2 (en) | 2008-04-11 | 2009-04-06 | Pesticidal compositions |
Country Status (21)
| Country | Link |
|---|---|
| US (5) | US8231887B2 (en) |
| EP (1) | EP2271216A4 (en) |
| JP (1) | JP2011517685A (en) |
| KR (1) | KR20100134105A (en) |
| CN (3) | CN103210959A (en) |
| AR (1) | AR071468A1 (en) |
| AU (1) | AU2009233944A1 (en) |
| BR (1) | BRPI0911664A2 (en) |
| CA (1) | CA2720953A1 (en) |
| CL (1) | CL2009000871A1 (en) |
| CR (1) | CR11720A (en) |
| EA (3) | EA201201029A1 (en) |
| IL (1) | IL208553A0 (en) |
| MA (1) | MA32292B1 (en) |
| MX (1) | MX2010011081A (en) |
| MY (1) | MY149026A (en) |
| PH (1) | PH12010502295A1 (en) |
| TW (1) | TW200948273A (en) |
| UY (1) | UY31759A (en) |
| WO (1) | WO2009126577A2 (en) |
| ZA (1) | ZA201008005B (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011062299A1 (en) * | 2009-11-20 | 2011-05-26 | Sumitomo Chemical Company, Limited | Pest control composition |
| WO2017108662A1 (en) * | 2015-12-22 | 2017-06-29 | Bayer Cropscience Aktiengesellschaft | Emulsion concentrates of lipophilic compounds |
| WO2019133423A1 (en) * | 2017-12-27 | 2019-07-04 | Mclaughlin Gormley King Company | Sabadilla alkaloid emulsifiable concentrate |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8231887B2 (en) | 2008-04-11 | 2012-07-31 | Basf Corporation | Pesticidal compositions |
| WO2011059054A1 (en) * | 2009-11-13 | 2011-05-19 | 国立大学法人 岡山大学 | Termite egg disinfestation technique utilizing volatile calling pheromone and queen pheromone |
| ITMI20110831A1 (en) * | 2011-05-12 | 2012-11-13 | Anna Andolfi | FAGODETERRENT COMPOUNDS OF MUSHROOM ORIGIN |
| KR101288541B1 (en) * | 2011-06-24 | 2013-07-22 | 전북대학교산학협력단 | Composition of acaricidal components containing Acetophenone's derivatives and acaricide using the same |
| US9999218B2 (en) | 2011-10-04 | 2018-06-19 | 0903608 B.C. Ltd. | Pest control formulations and methods of making and using same |
| CN108633932B (en) | 2011-10-04 | 2021-10-26 | 0903608 B.C.有限公司 | Pest control formulations and methods of making and using the same |
| CN114903848A (en) * | 2013-02-27 | 2022-08-16 | 阿根塔创新有限公司 | Transdermal preparation |
| AU2014238477B2 (en) | 2013-03-15 | 2017-04-06 | Keith Kennedy | Lice control composition and method |
| US9089141B2 (en) * | 2013-09-20 | 2015-07-28 | John James McIntyre | Natural insect repellent formula BBX2 |
| JP6603221B2 (en) * | 2013-12-30 | 2019-11-06 | ダウ グローバル テクノロジーズ エルエルシー | Bactericidal composition |
| EP3164374A4 (en) | 2014-07-02 | 2017-11-15 | Ralco Nutrition, Inc. | Agricultural compositions and applications utilizing essential oils |
| US20160374350A1 (en) * | 2015-06-29 | 2016-12-29 | Anthony C. Watson | Method of treating bed bug infestation and preventing transmission thereof |
| CN105794858A (en) * | 2016-03-01 | 2016-07-27 | 江西天人生物控股有限公司 | Farm-oriented insecticidal composition and application thereof |
| CN106035362A (en) * | 2016-05-31 | 2016-10-26 | 刘园 | Composition for controlling adoxophyes orana and preparation method of composition |
| CN108323525A (en) * | 2018-02-08 | 2018-07-27 | 青岛农业大学 | Application of the nerol in preparing termiticides |
| CN110122487B (en) * | 2019-05-23 | 2021-04-06 | 赣南师范大学 | Application of alcohol in attracting diaphorina citri and diaphorina citri attractant |
| TWI790635B (en) * | 2021-06-08 | 2023-01-21 | 誌懋股份有限公司 | Uses of Australian Cypress Extract for Cockroach Repellent |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001043547A1 (en) | 1999-12-16 | 2001-06-21 | Isp Investments Inc. | Fast drying biocidal preservative composition |
| US6251415B1 (en) | 1997-10-27 | 2001-06-26 | Fmc Corporation | Solvent system for pesticide products |
| US20040253287A1 (en) | 2002-08-03 | 2004-12-16 | Denton Robert Michael | Environmentally safe insecticides |
| US20050261379A1 (en) | 2004-05-18 | 2005-11-24 | Petro-Canada | Spray Oil and Method of Use Therefor for Controlling Turfgrass Pests |
| US20070020304A1 (en) | 2002-10-25 | 2007-01-25 | Foamix Ltd. | Non-flammable insecticide composition and uses thereof |
| WO2007047486A2 (en) | 2005-10-17 | 2007-04-26 | Yu Ruey J | Hydroxy-oligocarboxylic esters: effects on nerve and use for cutaneous and mucocutaneous organs or sites |
| US20070098750A1 (en) | 2005-09-01 | 2007-05-03 | Ecosmart Technologies, Inc. | Pesticidal compositions containing isopropyl myristate and analogs of same as a synergist for plant essential oils |
Family Cites Families (185)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US1516907A (en) * | 1918-06-25 | 1924-11-25 | Us Ind Alcohol Co | Fuel for motors |
| US1858635A (en) | 1927-04-25 | 1932-05-17 | Pan American Petroleum & Trans | Sweetening hydrocarbon oils |
| US2023140A (en) * | 1932-10-19 | 1935-12-03 | Shell Dev | Livestock spray |
| US2041874A (en) | 1935-02-15 | 1936-05-26 | Frits E Stockelbach | Composition for preventing sunburn |
| US2763614A (en) | 1954-06-04 | 1956-09-18 | Gulf Oil Corp | Corrosion-inhibiting compositions |
| US2809122A (en) * | 1954-06-24 | 1957-10-08 | Sherwin Williams Co | Dual purpose tinting base |
| BE581706A (en) * | 1959-08-14 | |||
| US3158535A (en) | 1961-10-02 | 1964-11-24 | Exxon Research Engineering Co | Pyrethrin-elastomer insecticidal composition and method |
| US3163607A (en) * | 1962-08-02 | 1964-12-29 | Bethlehem Steel Corp | Method for removing oil lubricant film from steel |
| US3291557A (en) * | 1963-01-07 | 1966-12-13 | Battelle Development Corp | Alkyl dicarboxylic acid treatment of leather |
| US3444080A (en) | 1967-04-14 | 1969-05-13 | Shell Oil Co | Lubricant for rolling metals |
| US4285868A (en) | 1968-03-20 | 1981-08-25 | Mobil Oil Corporation | Preparation of gamma-butyrolactones |
| US3923997A (en) | 1971-05-11 | 1975-12-02 | Rhodia | Process for repelling dogs and cats from a selected area or from each other using {65 -n-alkyl-{65 -butyrolactones and {67 -n-alkyl-{67 -valerolactones |
| GB1388836A (en) * | 1971-06-01 | 1975-03-26 | Medisan Ab | Cosmetic compositions |
| JPS5623967B2 (en) * | 1972-06-09 | 1981-06-03 | ||
| US4052353B1 (en) | 1974-01-02 | 1990-01-30 | Dispersions of water soluble polymers in oil | |
| US4056610A (en) | 1975-04-09 | 1977-11-01 | Minnesota Mining And Manufacturing Company | Microcapsule insecticide composition |
| US4279946A (en) * | 1977-09-07 | 1981-07-21 | Foseco International Limited | Coating compositions |
| JPS55167204A (en) * | 1979-06-15 | 1980-12-26 | Kao Corp | Composition for controlling agricultural and horticultural blight and noxious insect |
| US4252796A (en) | 1979-08-17 | 1981-02-24 | Yu Ruey J | Stable water-in-oil emulsions |
| US4562214A (en) | 1979-09-10 | 1985-12-31 | Witco Chemical Corporation | Personal care emulsion |
| AU548341B2 (en) * | 1982-02-02 | 1985-12-05 | Unilever Plc | Acne treatment composition |
| US4534128A (en) | 1983-02-01 | 1985-08-13 | Query Grady W | Method for pest control using anesthetic and insecticide |
| US4624070A (en) | 1983-02-01 | 1986-11-25 | Query Grady W | Method and composition for pest control using anesthetic and insecticide |
| US4933371A (en) * | 1984-07-26 | 1990-06-12 | Shirlo, Inc. | Controlling ticks and fleas with linalool |
| JPS6163603A (en) | 1984-09-04 | 1986-04-01 | Riken Koryo Kogyo Kk | Cockroach attractant |
| WO1986003940A1 (en) * | 1985-01-07 | 1986-07-17 | Rod Robert L | Reodorizing animal repellent process and product |
| US4923698A (en) * | 1986-12-19 | 1990-05-08 | S. C. Johnson & Son, Inc. | Bioactive film-forming composition for control of crawling insects and the like |
| US4822614A (en) | 1986-12-19 | 1989-04-18 | S. C. Johnson & Son, Inc. | Bioactive film-forming composition for control of crawling insects and the like |
| US4774082A (en) * | 1987-01-13 | 1988-09-27 | S. C. Johnson & Son, Inc. | Volatile insect repellents |
| US4774081A (en) | 1987-01-13 | 1988-09-27 | S. C. Johnson & Son, Inc. | Contact insect repellents |
| GB8723347D0 (en) * | 1987-10-05 | 1987-11-11 | Shell Int Research | Ectoparasiticidal pour-on formulation |
| US5510117A (en) | 1989-03-09 | 1996-04-23 | Micro-Pak, Inc. | Entrapment vehicle and method |
| GB8906382D0 (en) | 1989-03-20 | 1989-05-04 | Univ Southampton | Pheromone baits for social insects |
| US5143900A (en) * | 1989-05-19 | 1992-09-01 | Colgate-Palmolive Company | Perfumes containing N-lower alkyl neoalkanamide (s) |
| EP0400914B1 (en) | 1989-06-02 | 1996-06-26 | Pfizer Inc. | Agricultural foam compositions |
| US5093326A (en) | 1989-06-19 | 1992-03-03 | Stephen Herman | Repellant compositions |
| US4961532A (en) | 1989-07-07 | 1990-10-09 | Dow Corning Corporation | Fragrance release device containing a highly adsorptive copolymer |
| US5130135A (en) * | 1989-08-18 | 1992-07-14 | Smithkline Beecham Plc | Pesticidal formulations |
| US4962133A (en) | 1989-09-05 | 1990-10-09 | Dow Corning Corporation | Method of making highly adsorptive copolymers |
| US5290556A (en) * | 1990-02-02 | 1994-03-01 | The United States Of America As Represented By The Secretary Of Agriculture | Plastic bait composition for attracting and killing crop pests |
| US6316017B1 (en) | 1990-02-02 | 2001-11-13 | The United States Of America As Represented By The Secretary Of Agriculture | Composition and apparatus useful for attracting and controlling insect pests |
| US5227406A (en) * | 1990-04-09 | 1993-07-13 | Primavera Laboratories, Inc. | Insect repellent lotions and sprays |
| US5227163A (en) * | 1991-01-18 | 1993-07-13 | Clilco, Ltd. | Lice-repellant compositions |
| US5344776A (en) | 1991-03-28 | 1994-09-06 | The United States Of America As Represented By The Department Of Health And Human Services | DNA encoding an insect octopamine receptor |
| US5562864A (en) * | 1991-04-19 | 1996-10-08 | The Lubrizol Corporation | Lubricating compositions and concentrates |
| GB9115272D0 (en) | 1991-07-15 | 1991-08-28 | Pfizer Ltd | Benzimidazole anthelmintics |
| US5674518A (en) | 1991-08-06 | 1997-10-07 | Prentiss Incorporated | Method of fish management by poison fish bait method of making the bait, and formulation of bait |
| FR2689729B1 (en) | 1992-04-09 | 1994-06-03 | Roussel Uclaf | NOVEL PESTICIDE COMPOSITIONS CONTAINING A PYRETHRINOUIDE. |
| US5621013A (en) * | 1992-06-24 | 1997-04-15 | Primavera Laboratories, Inc. | Insect replellent blends, lotions, and sprays |
| MX9303636A (en) * | 1992-08-07 | 1994-07-29 | Us Agriculture | COMPOSITION AND METHOD TO REPELLENT ANTS. |
| US6001874A (en) * | 1992-08-18 | 1999-12-14 | Veierov; Dan | Arthropod control composition for plant protection |
| US5753686A (en) * | 1992-09-18 | 1998-05-19 | International Flavors & Fragrances Inc. | Method for repelling fire ants and horn flies and compositions for repelling fire ants and horn flies and acting as anti-feedants for fire ants and horn flies |
| JPH06116101A (en) * | 1992-10-05 | 1994-04-26 | Matsuda Michihiko | Spray composition for trapping small insect |
| US5653991A (en) * | 1993-03-24 | 1997-08-05 | The Rod Family Trust | Process for repelling and killing insects and compositions to effect the same comprising a monoterpene |
| WO1995000020A2 (en) * | 1993-06-17 | 1995-01-05 | Ecoscience Corporation | Method for control of conidial dispersal and improved maintenance of entomopathogenic fungal products |
| EP0646314B1 (en) | 1993-10-05 | 2004-08-11 | Sumitomo Chemical Company, Limited | Sustained release pesticidal or plant growth regulating composition |
| US5525597A (en) | 1993-12-14 | 1996-06-11 | Kalamazoo Holdings, Inc. | Synergistic insecticidal compositions comprising capsicum and insecticidal use thereof |
| US5914105A (en) * | 1994-06-17 | 1999-06-22 | Ecolab Inc. | Petroleum based pest bait |
| US5476609A (en) * | 1994-07-11 | 1995-12-19 | Wilkins, Jr.; William S. | Acidic cleaning composition for removing burnt starch from cold metal press heads |
| US5536501A (en) | 1994-12-30 | 1996-07-16 | Proguard, Inc. | Use of flavenoid aldehydes as insecticides and for killing arachnids |
| US5698210A (en) * | 1995-03-17 | 1997-12-16 | Lee County Mosquito Control District | Controlled delivery compositions and processes for treating organisms in a column of water or on land |
| US5679662A (en) | 1995-06-06 | 1997-10-21 | Thermo Trilogy Corporation | Synergistic use of azadirachtin and pyrethrum |
| US5559271A (en) | 1995-07-26 | 1996-09-24 | Phillips Petroleum Company | Organic polysulfide compositions having reduced odor |
| US5753507A (en) | 1995-09-22 | 1998-05-19 | Novartis Finance Corporation | Plant geraniol/nerol 10-hydroxylase and DNA coding therefor |
| US5654362A (en) | 1996-03-20 | 1997-08-05 | Dow Corning Corporation | Silicone oils and solvents thickened by silicone elastomers |
| US6277415B1 (en) | 1996-03-29 | 2001-08-21 | Orna Levin | Pediculocidal and veterinary compositions |
| WO1998000008A1 (en) | 1996-06-28 | 1998-01-08 | Novartis Ag | Pesticidal compositions |
| US5792465A (en) | 1996-06-28 | 1998-08-11 | S. C. Johnson & Son, Inc. | Microemulsion insect control compositions containing phenol |
| JP3855311B2 (en) | 1996-08-07 | 2006-12-06 | 住友化学株式会社 | Aerosol composition |
| US6184197B1 (en) | 1996-09-19 | 2001-02-06 | The Procter & Gamble Company | Polymeric compound comprising one or more active alcohols |
| US5834533A (en) * | 1996-11-20 | 1998-11-10 | Phillips Petroleum Company | Stable liquid suspension compositions |
| US5880210A (en) * | 1997-04-01 | 1999-03-09 | Dow Corning Corporation | Silicone fluids and solvents thickened with silicone elastomers |
| MA24577A1 (en) | 1997-06-26 | 1998-12-31 | Procter & Gamble | METHODS AND COMPOSITIONS FOR REDUCING MICROORGANISM FOR FOOD PRODUCTS |
| AU5679598A (en) | 1997-07-10 | 1999-02-08 | Earth Chemical Co. Ltd. | Pesticide applicator |
| DE19731156A1 (en) | 1997-07-21 | 1999-01-28 | Bayer Ag | Improved gel formulations containing insecticide |
| US5834413A (en) | 1997-09-26 | 1998-11-10 | Colgate-Palmolive Co. | Liquid cleaning compositions |
| US6074634A (en) | 1997-10-06 | 2000-06-13 | The United States Of America As Represented By The Secretary Of Agriculture | Feeding attractant and stimulant for adult control of noctuid and/or other lepidopteran species |
| EP1027288B1 (en) | 1997-10-14 | 2003-09-03 | Isp Investments Inc. | Stabilized concentrates of water unstable aza compounds and o/w miniemulsions thereof |
| AU1900899A (en) | 1997-10-27 | 1999-05-17 | Baylor College Of Medicine | Invertebrate octopamine receptor |
| US6130253A (en) | 1998-01-27 | 2000-10-10 | Ximed Group Plc | Terpene based pesticide treatments for killing terrestrial arthropods including, amongst others, lice, lice eggs, mites and ants |
| US5977186A (en) * | 1998-01-27 | 1999-11-02 | Ximed Group Plc | Terpene treatments for killing lice and lice eggs |
| DE19805703C2 (en) * | 1998-02-06 | 2001-05-03 | Cognis Deutschland Gmbh | Hair care products |
| US6083498A (en) * | 1998-03-10 | 2000-07-04 | The United States Of America As Represented By The Secretary Of Agriculture | Chemical attractants for yellowjackets and paper wasps |
| US6355264B1 (en) | 1998-03-20 | 2002-03-12 | Avon Products, Inc. | Insect repellent composition |
| US6706760B2 (en) | 1998-03-20 | 2004-03-16 | Sumitomo Chemical Company, Limited | Wasp controlling agent |
| US5998484A (en) | 1998-04-15 | 1999-12-07 | Woodstream Corporation | Insecticidal compositions and method of controlling insect pests using same |
| US6455086B1 (en) | 1998-06-26 | 2002-09-24 | The Procter & Gamble Company | Microorganism reduction methods and compositions for food cleaning |
| US6024972A (en) | 1998-07-21 | 2000-02-15 | Isp Investments Inc. | Water-free concentrate of amitraz insecticide and clear pour-on formulations thereof |
| AU2458599A (en) * | 1998-07-27 | 2000-02-21 | Carl J. Abraham | Vapor releasable, geraniols containing, tick and insect repellent compositions and methods |
| US20030194454A1 (en) | 1998-07-28 | 2003-10-16 | Bessette Steven M. | Pesticidal compositions containing rosemary oil and wintergreen oil |
| US6986898B1 (en) | 1999-06-28 | 2006-01-17 | Ecosmart Technologies, Inc. | Synergistic and residual pesticidal compositions containing plant essential oils with enzyme inhibitors |
| US6849614B1 (en) | 1998-07-28 | 2005-02-01 | Ecosmart Technologies, Inc. | Synergistic and residual pesticidal compositions containing plant essential oils |
| US6607716B1 (en) | 1998-09-29 | 2003-08-19 | Tech Labs, Inc. | Pediculicidal compositions, a kit, and methods of use |
| US6881776B2 (en) | 1998-10-29 | 2005-04-19 | Penreco | Gel compositions |
| ES2247834T3 (en) | 1998-10-29 | 2006-03-01 | Penreco | GEL COMPOSITIONS. |
| WO2000026285A1 (en) | 1998-10-29 | 2000-05-11 | Penreco | Gel compositions |
| EP1139735B1 (en) | 1998-12-24 | 2006-07-12 | University Of Southampton | Method and apparatus for dispersing a volatile composition |
| CA2372155A1 (en) | 1999-03-05 | 2000-09-08 | Ecosmart Technologies, Inc. | Herbicidal compositions containing plant essential oils and mixtures or blends thereof |
| US6531163B1 (en) * | 1999-06-28 | 2003-03-11 | Ecosmart Technologies, Inc. | Pesticidal compositions containing peppermint oil |
| WO2001000049A1 (en) | 1999-06-28 | 2001-01-04 | Ecosmart Technologies, Inc. | Pesticidal compositions containing plant essential oils against mites |
| US6949587B1 (en) | 1999-08-06 | 2005-09-27 | Ecosmart Technologies, Inc. | Pesticidal compositions containing plant essential oils against beetles |
| US6541517B1 (en) | 1999-09-03 | 2003-04-01 | Donald M. Murphy | Treatment of skin disorders |
| AU5912699A (en) | 1999-09-09 | 2001-04-10 | Ecosmart Technologies, Inc. | Pesticidal activity of plant essential oils and their constituents |
| JP4776757B2 (en) | 1999-10-01 | 2011-09-21 | 株式会社ダイゾー | Aerosol composition |
| US6124490A (en) | 1999-10-26 | 2000-09-26 | Mona Industries, Inc. | Zwitterionic siloxane polymers and ionically cross-linked polymers formed therefrom |
| ES2163999B1 (en) | 1999-11-11 | 2003-03-01 | Inabonos Sa | COMPOSITION OF NATURAL ORIGIN TO CONTROL THE POST-HARVEST PATHOLOGY OF FRUIT AND VEGETABLES AND APPLICATION METHOD. |
| US6448219B1 (en) | 2000-02-11 | 2002-09-10 | Iced Scents, Llc | Scented potpourri gel and method of making the same |
| WO2001060163A2 (en) | 2000-02-17 | 2001-08-23 | Ecosmart Technologies, Inc. | Plant essential oils cointaining pediculicidal compositions |
| WO2001072129A1 (en) | 2000-03-28 | 2001-10-04 | Council Of Scientific And Industrial Research | Acyclic monoterpenes as anti-sprouting agents for potato tubers |
| US6255356B1 (en) * | 2000-03-30 | 2001-07-03 | International Flavors & Fragrances Inc. | Method for inhibiting from feeding, cockroaches |
| US6514511B1 (en) | 2000-05-08 | 2003-02-04 | Beemaster, Inc. | Composition, method, and apparatus to attract bees |
| US6773700B2 (en) | 2000-05-08 | 2004-08-10 | Steven C. Thoenes | Composition, method, and apparatus to attract bees |
| US7282211B2 (en) | 2001-05-21 | 2007-10-16 | Btg International Inc. | Pest treatment composition |
| US20030060379A1 (en) | 2000-05-26 | 2003-03-27 | The Procter & Gamble Company | Pesticides |
| WO2001091560A2 (en) * | 2000-05-26 | 2001-12-06 | Ecosmart Technologies, Inc. | Pesticidal compositions containing mineral oil and/or soybean oil |
| US20030026823A1 (en) | 2000-06-05 | 2003-02-06 | The 54 Group, Ltd. | Environmentally safe insect repellent composition |
| US7081211B2 (en) | 2000-07-13 | 2006-07-25 | The Procter & Gamble Company | Multi-layer reaction mixtures and apparatuses for delivering a volatile component via a controlled exothermic reaction |
| US6673756B2 (en) | 2000-09-20 | 2004-01-06 | Symrise Gmbh & Co. Kg | Multiphase soaps |
| FR2816517B1 (en) | 2000-11-14 | 2002-12-27 | Agro Ind Rech S Et Dev Ard | PROCESS FOR THE PREPARATION OF SOLUBILIZATION ADJUVANTS FROM FUSEL OILS AND BONES |
| ES2242734T3 (en) | 2001-02-26 | 2005-11-16 | THE PROCTER & GAMBLE COMPANY | COMPOSITION TO RELEASE AROMA WITH IMPROVED STABILITY AND REDUCED FOAM FORMATION. |
| US6592637B2 (en) | 2001-03-16 | 2003-07-15 | Mcgee Thomas | Decorative candle and process for making same |
| CN1379988A (en) * | 2001-04-12 | 2002-11-20 | 丁一沅 | Environmental protection type plant source sanitary insecticide |
| US20020193346A1 (en) | 2001-04-26 | 2002-12-19 | Dairy Solutions, Llc. | Method and mixture for protecting animals against pests |
| US7820187B2 (en) | 2001-04-26 | 2010-10-26 | Dairy Solutions, Llc | Method and mixture for protecting animals against pests |
| US20050002980A1 (en) | 2001-06-29 | 2005-01-06 | Mullen Patricia A. | Fragrance-containing insect repellant compositions |
| US6749861B2 (en) | 2001-06-29 | 2004-06-15 | Lenco Laboratories, Llc | Fragrance-containing insect repellant compositions |
| WO2003020024A2 (en) | 2001-08-28 | 2003-03-13 | Eden Research Plc | Treatment and prevention of infections in plants |
| US6441051B1 (en) | 2001-09-20 | 2002-08-27 | William B. Wheeler | Insect eradicator and method |
| IL145767A (en) | 2001-10-04 | 2006-10-31 | Israel State | Microbicidal formulation comprising an essential oil or its derivatives |
| US20030165452A1 (en) | 2001-12-27 | 2003-09-04 | Avon Products, Inc. | Controlled released powder entrapment compositions for hydrophobic ingredient |
| US20030175369A1 (en) | 2002-02-19 | 2003-09-18 | Paragon Household Products Inc. | Natural moths repellent compositions |
| CN1481849A (en) | 2002-09-12 | 2004-03-17 | 胜 刘 | Oil essence for relieving fatigue and preserving health |
| US20060193789A1 (en) | 2002-10-25 | 2006-08-31 | Foamix Ltd. | Film forming foamable composition |
| US7622269B2 (en) | 2004-03-19 | 2009-11-24 | Tyratech, Inc. | Methods of screening tyramine- and octopamine-expressing cells for compounds and compositions having potential insect control activity |
| US20060263403A1 (en) | 2003-04-24 | 2006-11-23 | Essam Enan | Compositions and methods for controlling insects involving the tyramine receptor |
| BRPI0410491A8 (en) | 2003-04-24 | 2017-10-24 | Univ Vanderbilt | COMPOSITIONS AND METHODS FOR INSECT CONTROL |
| WO2004095926A2 (en) | 2003-04-28 | 2004-11-11 | Monsanto Technology, Llc | Treatment of plants and plant propagation materials with an antioxidant to improve plant health and/or yield |
| EP1504664A1 (en) | 2003-07-29 | 2005-02-09 | Novartis AG | Organic compounds |
| GB2405339A (en) | 2003-08-29 | 2005-03-02 | Reckitt Benckiser | Insecticidal stick composition |
| JP4393853B2 (en) * | 2003-12-11 | 2010-01-06 | 株式会社ナチュラルネットワーク | Cockroach spray |
| CN100355340C (en) * | 2003-12-16 | 2007-12-19 | 金中北 | Mineral oil microemulsion |
| CN1555699A (en) * | 2004-01-08 | 2004-12-22 | 金中北 | Aqueous emulsion green pesticide and its formula design and preparation method |
| US20050244445A1 (en) | 2004-04-15 | 2005-11-03 | Anderson David L | Insecticidal compositions and methods of using same |
| CN101084016A (en) | 2004-04-15 | 2007-12-05 | 克艾思马有限公司 | Compositions capable of facilitating penetration across a biological barrier |
| FR2869229B1 (en) | 2004-04-26 | 2006-08-25 | Sederma Soc Par Actions Simpli | USE OF A INDUCER OF UGT BY TOPIC |
| WO2006017232A1 (en) | 2004-07-12 | 2006-02-16 | Taro Pharmaceuticals Industries Ltd. | Topical gel formulation comprising insecticide and its preparation thereof |
| US7531187B2 (en) | 2004-07-13 | 2009-05-12 | United Phosphorus, Ltd. | Synergistic insecticidal composition containing chloronicotynyle and pyrethroids compounds |
| US20060034898A1 (en) | 2004-08-10 | 2006-02-16 | Amodt James A | Method for using FIFRA-exempt substances, materials exhibiting antimicrobial, insecticidal, biocidal and/or odor controlling properties having one or more of these substances coated thereon and/or incorporated therein, and products that are made using these materials |
| GB0418047D0 (en) | 2004-08-12 | 2004-09-15 | Syngenta Participations Ag | Fungicidal compositions |
| GB0422400D0 (en) | 2004-10-08 | 2004-11-10 | Syngenta Participations Ag | Fungicidal compositions |
| GB0422401D0 (en) | 2004-10-08 | 2004-11-10 | Syngenta Participations Ag | Fungicidal compositions |
| US20060083763A1 (en) | 2004-10-18 | 2006-04-20 | Michael Neale | Method and apparatus for insect repellant system |
| US20060116353A1 (en) | 2004-11-26 | 2006-06-01 | Arthur Vanmoor | Method for removal of unwanted organism from the human body and a treatment for removal of unwanted organism from the human body |
| US20060211575A1 (en) | 2005-03-16 | 2006-09-21 | W. Neudorff Gmbh Kg | Control for plant and plant product pathogens |
| US20060222675A1 (en) | 2005-03-29 | 2006-10-05 | Sabnis Ram W | Personal care compositions with color changing indicator |
| US20060257441A1 (en) | 2005-05-10 | 2006-11-16 | Suzuki Yushi Industrial Co., Ltd. | Natural essential oil sanitary insect pest repellent and its use |
| DE102005026801A1 (en) | 2005-06-10 | 2006-12-14 | Symrise Gmbh & Co. Kg | sandalwood odoriferous |
| US8062676B2 (en) | 2005-08-02 | 2011-11-22 | Thomas Besendorfer | Pesticide composition |
| US8007820B2 (en) | 2005-09-16 | 2011-08-30 | W. Neudorff Gmbh Kg | Wide spectrum insecticide and miticide composition |
| GB0522287D0 (en) | 2005-11-01 | 2005-12-07 | Reckitt Benckiser Uk Ltd | Method and compositions |
| WO2007056340A2 (en) | 2005-11-08 | 2007-05-18 | Mycosol, Inc. | Disinfectant and antiseptic pyridinium and thiazolium compounds and methods of using the same |
| WO2007068998A1 (en) | 2005-12-15 | 2007-06-21 | Sederma | New polypeptides and their use |
| US20070178128A1 (en) | 2006-01-06 | 2007-08-02 | Ecosmart Technologies, Inc. | Emulsifiable concentrate (ec) formulations for pesticides |
| US20070190094A1 (en) | 2006-01-10 | 2007-08-16 | Ecosmart Technologies, Inc. | Pesticidal compositions containing isopropyl-containing compounds as pesticidally active ingredients |
| US7846464B2 (en) | 2006-01-16 | 2010-12-07 | Darling Samuel T | Insect repellent composition |
| US8404648B2 (en) | 2006-02-16 | 2013-03-26 | Sederma | Polypeptides KXK and their use |
| EP2090662A3 (en) | 2006-04-05 | 2012-10-31 | Metanomics GmbH | Process for the production of a fine chemical |
| CA2650596A1 (en) | 2006-04-28 | 2007-11-08 | Buckman Laboratories International, Inc. | Fungicidal compositions and methods of using the same |
| WO2007146114A2 (en) | 2006-06-08 | 2007-12-21 | Tyratech, Inc | Compositions and methods for production of peat-like material from manure |
| US7824668B2 (en) | 2006-07-12 | 2010-11-02 | Cotton Incorporated | Compositions for attracting stink bugs |
| JP2009543870A (en) | 2006-07-17 | 2009-12-10 | タイラテック, インク. | Compositions and methods for insect control |
| DE102006044849A1 (en) | 2006-09-22 | 2008-04-03 | Symrise Gmbh & Co. Kg | Deconjugated alkadienoic esters as fragrances and / or flavorings |
| US8293286B2 (en) | 2006-10-16 | 2012-10-23 | Sergeant's Pet Care Products, Inc. | Natural compositions for killing parasites on a companion animal |
| WO2008056365A2 (en) | 2006-11-09 | 2008-05-15 | Y.A.D. Bio-Agrotech Ventures Ltd. | Pediculicidal, pesticidal or insecticidal compositions |
| US8758787B2 (en) | 2006-12-04 | 2014-06-24 | Fasst Products, LLC. | Long-lasting insect repellant, pesticide and antifeedant compositions |
| US20080159970A1 (en) | 2006-12-20 | 2008-07-03 | L'oreal | Kit comprising silicone compounds and a cosmetic and/or dermatological active agent |
| BRPI0806666A8 (en) | 2007-01-16 | 2018-02-06 | Tyratech Inc | COMPOSITIONS AND METHOD FOR PEST CONTROL |
| US20080187607A1 (en) | 2007-02-01 | 2008-08-07 | Ecosmart Technologies Inc. | Systems, methods and apparatuses for epa exempt pesticidal compositions |
| US20080193387A1 (en) | 2007-02-14 | 2008-08-14 | Ricki De Wolff | Essential oil compositions for killing or repelling ectoparasites and pests and methods for use thereof |
| US9049814B2 (en) | 2007-02-23 | 2015-06-09 | Vamtech, Llc | Coated seeds and methods of making coated seeds |
| JP5326320B2 (en) | 2007-03-30 | 2013-10-30 | 住友化学株式会社 | Pest control composition and pest control method |
| US7588646B2 (en) | 2007-05-03 | 2009-09-15 | United Laboratories, Inc. | Cleaning compositions containing an alkyl ester and methods of using same |
| WO2009012415A1 (en) | 2007-07-17 | 2009-01-22 | Tyratech, Inc. | Insect repellent composition |
| US8231887B2 (en) * | 2008-04-11 | 2012-07-31 | Basf Corporation | Pesticidal compositions |
-
2009
- 2009-04-03 US US12/418,443 patent/US8231887B2/en not_active Expired - Fee Related
- 2009-04-03 US US12/418,455 patent/US9005644B2/en active Active
- 2009-04-03 US US12/418,466 patent/US20090258889A1/en not_active Abandoned
- 2009-04-06 CN CN2013101327181A patent/CN103210959A/en active Pending
- 2009-04-06 MX MX2010011081A patent/MX2010011081A/en active IP Right Grant
- 2009-04-06 WO PCT/US2009/039664 patent/WO2009126577A2/en active Application Filing
- 2009-04-06 PH PH12010502295A patent/PH12010502295A1/en unknown
- 2009-04-06 JP JP2011504111A patent/JP2011517685A/en active Pending
- 2009-04-06 BR BRPI0911664-8A patent/BRPI0911664A2/en not_active IP Right Cessation
- 2009-04-06 EP EP09730159A patent/EP2271216A4/en not_active Withdrawn
- 2009-04-06 MY MYPI20104721 patent/MY149026A/en unknown
- 2009-04-06 EA EA201201029A patent/EA201201029A1/en unknown
- 2009-04-06 AU AU2009233944A patent/AU2009233944A1/en not_active Abandoned
- 2009-04-06 EA EA201001616A patent/EA018969B1/en not_active IP Right Cessation
- 2009-04-06 EA EA201201028A patent/EA201201028A1/en unknown
- 2009-04-06 CN CN2013101326390A patent/CN103190446A/en active Pending
- 2009-04-06 CA CA2720953A patent/CA2720953A1/en not_active Abandoned
- 2009-04-06 CN CN2009801215262A patent/CN102083311A/en active Pending
- 2009-04-06 KR KR20107025258A patent/KR20100134105A/en not_active Application Discontinuation
- 2009-04-08 AR ARP090101274 patent/AR071468A1/en not_active Application Discontinuation
- 2009-04-09 CL CL2009000871A patent/CL2009000871A1/en unknown
- 2009-04-10 TW TW98112120A patent/TW200948273A/en unknown
- 2009-04-13 UY UY31759A patent/UY31759A/en unknown
-
2010
- 2010-10-07 IL IL208553A patent/IL208553A0/en unknown
- 2010-10-08 CR CR11720A patent/CR11720A/en not_active Application Discontinuation
- 2010-11-08 MA MA33331A patent/MA32292B1/en unknown
- 2010-11-09 ZA ZA2010/08005A patent/ZA201008005B/en unknown
-
2012
- 2012-07-30 US US13/562,073 patent/US8591927B2/en active Active
-
2013
- 2013-05-08 US US13/889,896 patent/US20130245053A1/en not_active Abandoned
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6251415B1 (en) | 1997-10-27 | 2001-06-26 | Fmc Corporation | Solvent system for pesticide products |
| WO2001043547A1 (en) | 1999-12-16 | 2001-06-21 | Isp Investments Inc. | Fast drying biocidal preservative composition |
| US20040253287A1 (en) | 2002-08-03 | 2004-12-16 | Denton Robert Michael | Environmentally safe insecticides |
| US20070020304A1 (en) | 2002-10-25 | 2007-01-25 | Foamix Ltd. | Non-flammable insecticide composition and uses thereof |
| US20050261379A1 (en) | 2004-05-18 | 2005-11-24 | Petro-Canada | Spray Oil and Method of Use Therefor for Controlling Turfgrass Pests |
| US20070098750A1 (en) | 2005-09-01 | 2007-05-03 | Ecosmart Technologies, Inc. | Pesticidal compositions containing isopropyl myristate and analogs of same as a synergist for plant essential oils |
| WO2007047486A2 (en) | 2005-10-17 | 2007-04-26 | Yu Ruey J | Hydroxy-oligocarboxylic esters: effects on nerve and use for cutaneous and mucocutaneous organs or sites |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2271216A4 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011062299A1 (en) * | 2009-11-20 | 2011-05-26 | Sumitomo Chemical Company, Limited | Pest control composition |
| US9265256B2 (en) | 2009-11-20 | 2016-02-23 | Sumitomo Chemical Company, Limited | Pest control composition |
| WO2017108662A1 (en) * | 2015-12-22 | 2017-06-29 | Bayer Cropscience Aktiengesellschaft | Emulsion concentrates of lipophilic compounds |
| US11369108B2 (en) | 2015-12-22 | 2022-06-28 | Bayer Cropscience Aktiengesellschaft | Emulsion concentrates of lipophilic compounds |
| WO2019133423A1 (en) * | 2017-12-27 | 2019-07-04 | Mclaughlin Gormley King Company | Sabadilla alkaloid emulsifiable concentrate |
| US10874099B2 (en) | 2017-12-27 | 2020-12-29 | Mclaughlin Gormley King Company | Sabadilla alkaloid emulsifiable concentrate |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8591927B2 (en) | Pesticidal compositions | |
| EP1855527B1 (en) | Limonene-containing herbicide compositions | |
| Ciriminna et al. | Herbicides based on pelargonic acid: Herbicides of the bioeconomy | |
| US8993004B2 (en) | Pest control composition | |
| US20160262404A1 (en) | Pesticide Compositions Containing Thyme Oil and Cinnamon Oil | |
| US20220354127A1 (en) | Insect, bacterial, and/or fungal control composition | |
| US20230354824A1 (en) | Minimum Risk Pesticide Formulations Containing Mineral Oil | |
| US20230354825A1 (en) | Water-Based Pesticide Formulations For Yard Applications | |
| Ferreira et al. | Chemistry of sex pheromones and their role in integrated pest management | |
| AU2021318836A1 (en) | Insect control composition | |
| WO2019086799A1 (en) | Pesticide or repellent based on geraniol or citronellal |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980121526.2 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09730159 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2720953 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011504111 Country of ref document: JP Ref document number: 201011720 Country of ref document: CR Ref document number: 12010502295 Country of ref document: PH Ref document number: MX/A/2010/011081 Country of ref document: MX Ref document number: CR2010-011720 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009730159 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009233944 Country of ref document: AU Ref document number: 3855/KOLNP/2010 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201001616 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: 20107025258 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2009233944 Country of ref document: AU Date of ref document: 20090406 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: PI0911664 Country of ref document: BR Kind code of ref document: A2 Effective date: 20101011 |