PHARMACEUTICAL FORMULATION
An oral film coated tablet composition containing the antibiotic Cefuroxime Axetil in the core and a specially designed double layered film coating which serves to delay the film rupture time to avoid potential bitter taste due to rapid film rupture of tablet upon oral administration.
This invention is concerned with pharmaceutical compositions containing the 1-acetoxyethyl ester of Cefuroxime which has an approved name, 'Cefuroxime Axetil'.
Cefuroxime, as disclosed in British Patent Specification No. 1453049, is a valuable broad spectrum antibiotic having high activity against a wide range of gram positive and gram negative microorganisms. Cefuroxime and its salts are principally of value as injectable antibiotics, since they are poorly absorbed from the gastro-intestinal tract. It has been disclosed in US Patent specification No. 4897270, that esterification of the carboxyl group of Cefuroxime as a 1-acetoxyethylester to give Cefuroxime Axetil improves the effectiveness on oral administration as disclosed in British Patent Specification No. 1571683. The presence of the 1-acetoxyethyl esterifying group results in significant absorption of the compound from the gastro-intestinal tract, whereupon the esterifying group is hydrolysed by enzymes present in, for example, in serum and body tissues to yield the antibiotically active acid. Employing Cefuroxime Axetil in an amorphous form as disclosed in British Patent Specification No.2127401, is also found to be advantageous. Cefuroxime Axetil has therefore extended the valuable therapeutic potential of Cefuroxime by making available a form of the antibiotic which may be administered orally rather than by injection only. Cefuroxime Axetil is necessarily presented as a tablet for oral administration, as this is the most convenient method. The long-lasting
extremely bitter taste of Cefuroxime Axetil, is a real challenge, since this bitter taste cannot be adequately masked by addition of sweeteners and flavors. Taste masking of drugs through the use of flavors is well known. However, flavoring ingredients are not sufficient to mask certain drugs. Cefuroxime is one such drug, which has an extremely bitter taste which is long lasting and which cannot be masked by the addition of sweeteners and flavors. Again, since one of the most convenient means of presenting Cefuroxime Axetil for oral administration, being a tablet, it has been found necessary to use tablets that are film coated to mask the bitter taste.
Conventionally coated Cefuroxime Axetil tablets, complied with the standard disintegration tests (with discs) specified in the British Pharmacopoeia [1998, Appendix XII A, pg. A1871 and United States Pharmacopoeia [USP-24, pg. 1941]. However, it was found that when such film coated tablets were administered to human volunteers low levels of absorption of Cefuroxime Axetil were obtained from the gastro-intestinal tract. It has been reported in the art, as disclosed in US Patent No. 4897270, that Cefuroxime Axetil once in contact with aqueous media can form a gelatinous mass. This gelling effect is temperature dependent, but does occur at temperatures of about 37°C, i.e. at the physiological temperatures at which the disintegration of orally administered tablets takes place. Further, with the relatively slow penetration of moisture, from the film coat to the core, which occurs upon administration of tablets of Cefuroxime Axetil provided with conventional coats, the Cefuroxime Axetil present in the tablet core may gel. This gel formation leads to poor dissolution of Cefuroxime Axetil; thus the absorption from the gastro-intestinal tract is greatly reduced. This occurs both with the crystalline and amorphous form of Cefuroxime Axetil. This problem of gelling has been overcome in the
US Patent No. 4897270, by employing a film coating that ruptures rapidly, upon contact with the gastrointestinal fluid, and the core disintegrates immediately, thus allowing dispersion and dissolution of the Cefuroxime Axetil in the gastro-intestinal tract before any gelling effect can occur, thus ensuring high bioavailability of Cefuroxime Axetil.
However, in the composition outlined in US Patent No. 4897270, the rupture time for the tablet is clocked at less than 40 sees, as claimed in the patent. The patent has further claimed the rupture time to be less than 25 sees or less that 15 sees. This rapid rupture can be considered as a disadvantage, especially in case of patients who are not very comfortable in swallowing oral medications like tablets and capsules. In general swallowing a tablet is considered very difficult by most of the people. Some people take a swig of water/other swallowing media viz. milk, place the tablet also into the mouth and gulp down the tablet along with the swallowing media. There are still others who first place the tablet on the tongue, then take a swig of the swallowing media and then gulp down the tablet. In spite of this, the inherent fear of not swallowing the tablet at the first try still persists. This one can observe in case of normal adults. In case of children and old people, who find difficulty swallowing their regular food, swallowing a tablet requires tremendous effort. Effectively, in case of patients who are in inherent fear of swallowing oral medications or in case of those who have difficulty swallowing, the time taken to gulp down the tablet after it is placed in the swallowing media, can possibly be more than 40 sees. If the patient takes slightly longer time to swallow the tablet, due to inability of swallowing the tablet as in the case of old people or children, or in case of other patients who are not able to swallow the tablet in the first try due to inherent fear and hence end up placing the tablet in the
swallowing media more than once, it effectively takes more than 40 sees to swallow the tablet. The very purpose of masking the bitter taste of the Cefuroxime Axetil, in the core of the tablet, by using a film coat, stands defeated by employing a rupture coat, which ruptures below 40 sees.
The present invention aims at providing a solution for this problem, by prolonging the rupture time of the film coat beyond 40 seconds. The design of the film, ensures the rupture time in the range between 45-240 seconds, after which, the core immediately disintegrates and gives better dissolution, ultimately yielding better bioavailability. With reference to the present invention, to obtain film coats which delay film rupture time beyond 45 seconds but less than 240 seconds, it is preferred to apply a thin first coat of film- forming composition comprising of HPMC (5-50 cps) uniformly onto the tablet core to serve the purpose of masking the bitter taste of the drug, followed by a second thin coat of a water insoluble polymer e.g. Shellac, to delay the film rupture time beyond 45 seconds.
Further, to obtain tablet cores which disintegrate quickly, i.e. within 1 minute, following rupture of film coat in the rupture test, it is necessary to incorporate into the core an effective disintegrant in an effective amount.
Film forming agents which are useful, include, hydroxypropyl cellulose, hydroxypropyl methylcellulose such as 5, 6 or 15 cps or hydroxypropyl methylcellulose 50 cps. Which may be used as aqueous or non-aqueous solvent system on the tablet core to mask the bitter taste of the drug Cefuroxime Axetil. Again as disclosed in US Patent No. 4865851, any holes in the coating would mean that the bitter taste was not effectively masked. Rapid rupture of film creates holes on the tablet surface and the drug may give bitter taste in the mouth itself, before it is swallowed.
In other cases, where an effort to swallow the tablet at the first go fails, and the tablet is taken in the mouth for a second time, there may be holes created in the first try, due to the rapid rupture property of the film coat.
According to another embodiment of the present invention, a second coat of water insoluble polymer, Shellac, in suitable concentration is applied over the tablet previously coated with a film coat of HPMC. The Shellac layer is coated to delay the film rupture time. The shellac used is dewaxed shellac, refined shellac or bleached shellac, dissolved in suitable solvent system.
As per another embodiment of the present invention, the weights of the film coat applied to tablets according to the invention is preferably in the range of 2-5% weight gain of first HPMC film coat and 0.1 to 1.0% weight gain of second coat of Shellac polymer.
According to other embodiment of the present invention, the tablet core may be formulated such that it disintegrates within 1 minute following rupture of the film coat, using the methods well known in the pharmaceutical art. This may usually be achieved by using disintegrants like dried starch, sodium starch glycolate, crosspovidone and cross linked carboxymethyl cellulose sodium (crosscarmellose sodium). The tablet core may comprise from 2-15% by weight of disintegrant. Other excipients which may be present in the tablet core of the film coated tablet of the invention are, Binding agents e.g. pregelatinised maize starch, polyvinyl pyrrolidone or hydroxypropyl methyl cellulose; Filler e.g. starch, lactose, microcrystalline cellulose and dicalcium phosphate; Lubricants and flow aids e.g. talc, aerosil, stearic acid, magnesium stearate preferably hydrogenated castor oil and Wetting agents e.g. sodium lauryl sulphate.
From the foregoing description, one of ordinary skill in the art can easily ascertain the essential characteristics of this invention, and without departing from the spirit and scope thereof, can make various changes and modifications of the invention to adapt it to various usages and conditions. As such, such changes and modifications are properly, equitably, and intended to be, within the full range of equivalence of the following claims. The following examples illustrate the invention in further detail.
EXPERIMENTS
Manufacturing procedure : All the ingredients excluding aerosil and half the quantity of croscarmellose sodium were blended together and compacted using a suitable roller compactor or slugged, the flakes or slugged material was comminuted using an oscillating granulator or multimilled through to get suitable granules. The resultant granules were blended with remaining excipients and then compressed to tablets using suitable compression machine meeting physical parameters such as hardness, friability, thickness, weight variation and disintegration tests.
Coating formula :
The film forming composition was prepared by dispersing the ingredients in solvent mixture. It was then applied by spray coating over 20,000 tablets in a 12" ganscoater with a target weight gain of 2 to 2.5%.
The shellac is dissolved in solvent and filtered. The filtered shellac solution was sprayed over previously HPMC coated tablet uniformly in a 12" ganscoater with a target weight gain of 0.1 to 0.5%. Mean film coat rupture time (20 Tablets) 45 seconds to 120 seconds.
Example 2 : 250 mg strength.
Core tablet composition :
Manufacturing procedure : All the ingredients excluding aerosil and half the quantity of croscarmellose sodium were blended together and compacted using a suitable roller compactor or slugged, the flakes or slugged material was comminuted using an oscillating granulator or multimilled through to get suitable granules. The resultant granules were blended with remaining excipients and then compressed to tablets using suitable compression machine meeting to physical parameters such as hardness, friability, thickness, weight variation and disintegration tests.
Coating formula :
The film forming composition was prepared by dispersing the ingredients in solvent mixture. It was then applied by spray coating over 20,000 tablets in a 12" ganscoater with a target weight gain of 2 to 2.5%.
The shellac is dissolved in solvent and filtered. The filtered shellac solution was sprayed over previously HPMC coated tablet uniformly in a 12" ganscoater with a target weight gain of 0.1 to 0.5%.
Mean film coat rupture time (20 Tablets) 45 seconds to 120 seconds.
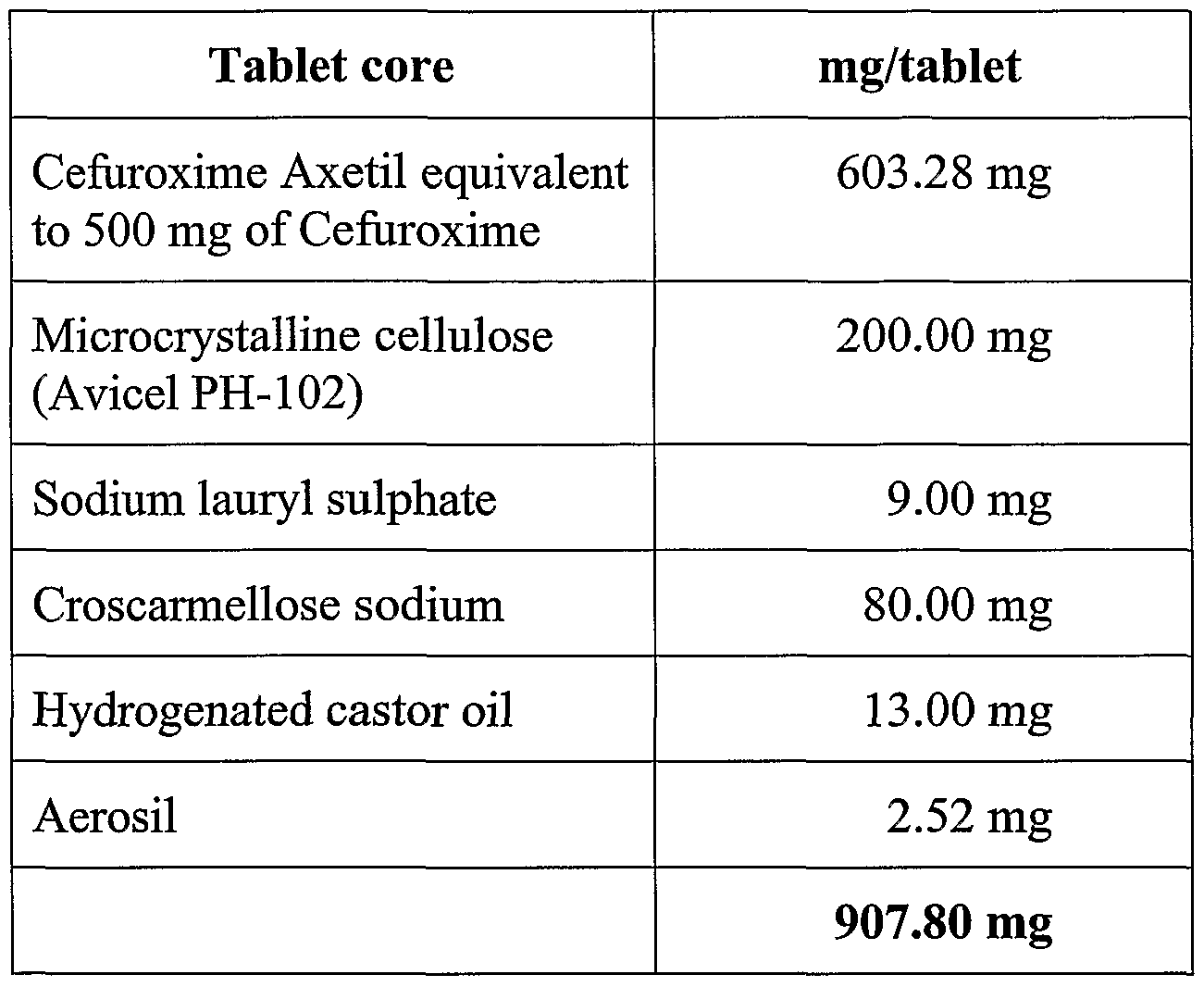
Example 3 : 500 mg strength.
Core tablet composition :
Manufacturing procedure : All the ingredients excluding aerosil and half the quantity of croscarmellose sodium were blended together and compacted using a suitable roller compactor or slugged, the flakes or slugged material was comminuted using an oscillating granulator or multimilled through to get suitable granules. The resultant granules were blended with remaining excipients and then compressed to tablets using suitable compression machine meeting to physical parameters such as hardness, friability, thickness, weight variation and disintegration tests.
Coating formula
The film forming composition was prepared by dispersing the ingredients in solvent mixture. It was then applied by spray coating over 20,000 tablets in a 12" ganscoater with a target weight gain of 2 to 2.5%.
The shellac is dissolved in solvent and filtered. The filtered shellac solution was sprayed over previously HPMC coated tablet uniformly in a 12" ganscoater with a target weight gain of 0.1 to 0.5%. Mean film coat rupture time (20 Tablets) 45 seconds to 120 seconds.
The examples quoted above, should not be construed to limit the scope of the invention.